Team:Tokyo Tech/Project/Artificial Cooperation System/lux act rep/Pluxrep/assay1
From 2010.igem.org
(→Materials & Methods) |
|||
| (7 intermediate revisions not shown) | |||
| Line 21: | Line 21: | ||
We confirmed that 3OC6HSL repressed luxR repression promoter, R0061, as expected. | We confirmed that 3OC6HSL repressed luxR repression promoter, R0061, as expected. | ||
===Materials & Methods=== | ===Materials & Methods=== | ||
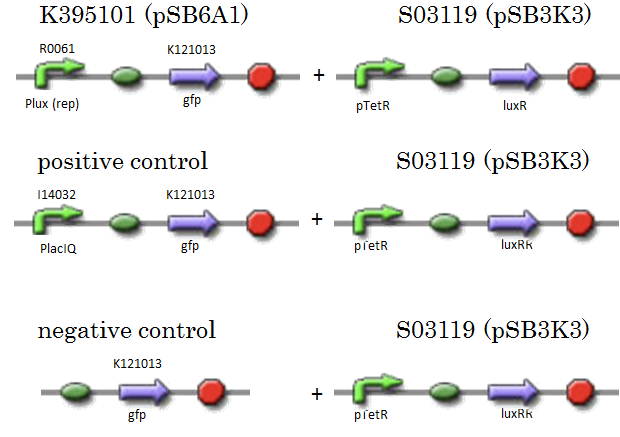
| - | We constructed K395101 combining R0061 and K121013 | + | We constructed K395101 combining R0061 and K121013, which is a promoter-less gfp reporter (rbs-gfp-ter-ter) on pSB6A1. S03119 is a LuxR generator which is repressed by TetR. In this experiment, we don’t use TetR, therefore, S03119 functions a LuxR constitutive generator. The backbone of S03119 is pSB1A2, which is a high copy plasmid, so we changed the backbone from pSB1A2 to pSB3K3. |
We used a fusion of PlacI<sup>q</sup> (I14032) to gfp (K121013) as a positive control and used promoterless gfp (K121013) as a negative control. | We used a fusion of PlacI<sup>q</sup> (I14032) to gfp (K121013) as a positive control and used promoterless gfp (K121013) as a negative control. | ||
| - | |||
| - | + | [[IMAGE:Tokyotech_R0061assay_construction.png|400px]] | |
| - | + | ||
| - | + | ||
| - | + | *'''samples''' | |
| - | + | #[Plux rep - GFP](BBa_K395101) on pSB6A1 + [PtetR – LuxR] on pSB3K3 | |
| + | #positive control: [PlacI<sup>q</sup>(constitutive promoter) - GFP] on pSB6A1+ [PtetR – LuxR] on pSB3K3 | ||
| + | #negative control: [promoterless - GFP] on pSB6A1+ [PtetR – LuxR] on pSB3K3 | ||
| + | *'''Strain''' | ||
DH5&alpha | DH5&alpha | ||
| - | + | *'''protocol''' | |
| - | + | #Prepare overnight culture. | |
| - | + | #Take 30 ul of the overnight culture into LB + antibiotics (Amp + Kan).(→fresh culture) | |
| - | (→fresh culture) | + | #Incubate the fresh culture until the observed O.D. reaches around 0.80. |
| - | + | #Each sample was divided into 2. Prepare and add 3OC6HSL mixture to one, and add DMSO mixture to the other. The final concentration of AHL is 100nM. | |
| - | + | #Induction for 2 hours at 37°C. | |
| - | Prepare and add 3OC6HSL mixture to one, and add DMSO mixture to the other. | + | #Fluorometer (FLA5200) and flow cytometry measurements for GFP expression. |
| - | The final concentration of AHL is 100nM. | + | |
| - | + | ||
| - | + | ||
===Reference=== | ===Reference=== | ||
| - | + | #KRISTI A. EGLAND & E. P. GREENBERG Conversion of the Vibrio fischeri Transcriptional Activator, LuxR, to a Repressor JOURNAL OF BACTERIOLOGY, Feb. 2000, p. 805–811 | |
Latest revision as of 14:36, 26 October 2010
Contents |
luxR repression promoter (BBa_R0061) assay
Abstract
We characterized the strength of this promoter which has never been done before in BioBrick in order to design new promoter based on this data. We measured fluorescence by flow cytometry 3 hour after addition of 100nM 3OC6HSL to confirm R0061, which is an existing BioBrick promoter repressed by LuxR/3OC6HSL complex.
Introduction
We characterized the strength of this promoter which has never been done before in BioBrick in order to design new promoter base on this data. In the Artificial Cooperation System, we inserted chloramphenicol resistance coding sequence to this promoter. Thus, this promoter plays an important role in Artificial Cooperation System.
Results
The result is shown in fig.○. The expression of GFP with 3OC6HSL dropped to 1/3 comparing with the expression without 3OC6HSL.
Conclusion
We confirmed that 3OC6HSL repressed luxR repression promoter, R0061, as expected.
Materials & Methods
We constructed K395101 combining R0061 and K121013, which is a promoter-less gfp reporter (rbs-gfp-ter-ter) on pSB6A1. S03119 is a LuxR generator which is repressed by TetR. In this experiment, we don’t use TetR, therefore, S03119 functions a LuxR constitutive generator. The backbone of S03119 is pSB1A2, which is a high copy plasmid, so we changed the backbone from pSB1A2 to pSB3K3. We used a fusion of PlacIq (I14032) to gfp (K121013) as a positive control and used promoterless gfp (K121013) as a negative control.
- samples
- [Plux rep - GFP](BBa_K395101) on pSB6A1 + [PtetR – LuxR] on pSB3K3
- positive control: [PlacIq(constitutive promoter) - GFP] on pSB6A1+ [PtetR – LuxR] on pSB3K3
- negative control: [promoterless - GFP] on pSB6A1+ [PtetR – LuxR] on pSB3K3
- Strain
DH5&alpha
- protocol
- Prepare overnight culture.
- Take 30 ul of the overnight culture into LB + antibiotics (Amp + Kan).(→fresh culture)
- Incubate the fresh culture until the observed O.D. reaches around 0.80.
- Each sample was divided into 2. Prepare and add 3OC6HSL mixture to one, and add DMSO mixture to the other. The final concentration of AHL is 100nM.
- Induction for 2 hours at 37°C.
- Fluorometer (FLA5200) and flow cytometry measurements for GFP expression.
Reference
- KRISTI A. EGLAND & E. P. GREENBERG Conversion of the Vibrio fischeri Transcriptional Activator, LuxR, to a Repressor JOURNAL OF BACTERIOLOGY, Feb. 2000, p. 805–811
 "
"