Team:Korea U Seoul/Notebook
From 2010.igem.org
| Line 26: | Line 26: | ||
</style> | </style> | ||
</html> | </html> | ||
| - | + | ||
{| align="center" | {| align="center" | ||
|-valign="top" | |-valign="top" | ||
| Line 35: | Line 35: | ||
|align="center" width="150pt"|{{#calendar: title=Korea_U_Seoul |year=2010 | month=10}} | |align="center" width="150pt"|{{#calendar: title=Korea_U_Seoul |year=2010 | month=10}} | ||
|} | |} | ||
| - | + | <div id="yong"> | |
== Experimental notes == | == Experimental notes == | ||
Revision as of 13:10, 27 October 2010

Brain storming & Work notes
Click on a date to see notes on the meeting & summary of labwork done on that day.
|
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Experimental notes
[Discussion] 2010-08-02 ~ 2010-08-29
1. Strategy and overview of iGEM 2010 experiment
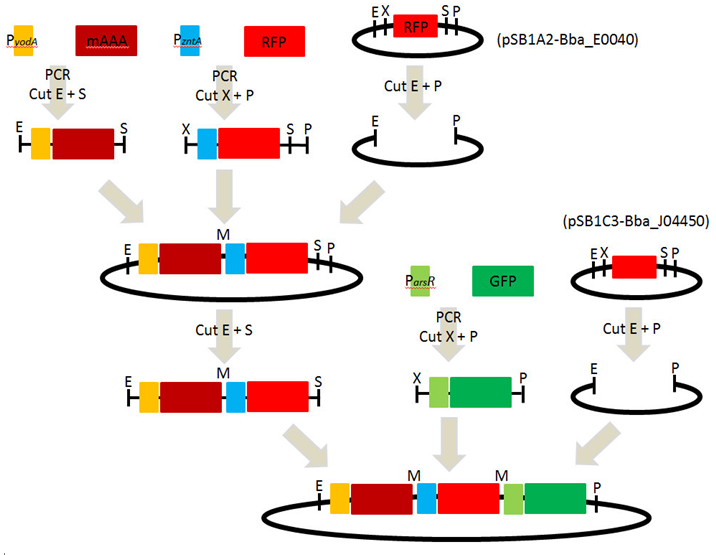
2. Design of primers
| Primer | Sequence ( 5’ → 3’ ) |
|---|---|
| PyodA(EcoRI)_F | zz |
| mAAA(SpeI)_R | zzzz |
| PzntA(XbaI)_F | xzzzz |
| RFP(PstI)_R | cxvcxv |
| ParsR(XbaI)_F | zzzz |
| GFP(PstI)_R | asasas |
[ Preparation of competent cells ] 2010-09-01 ~ 2010-09-03
1. Inoculation of E. coli DH5α and E. coli BL21(DE3) to 3mL LB broth
2. Preparation of 200mL 2x LB broth, TSS solution and LB plates with ampicillin(100μg/mL) and chloramphenicol(25μg /mL), respectively
3. Inoculation of subcultured E. coli to 200mL 2x LB borth
4. Preparation of competent cells by CSBL laboratory protocol
5. Transformation of pUC19 plasmid(10ng/μL) to competent cells for transformation efficiency check
[ Transformation efficiency ] 2010-09-04
| Strain | Number of colonies (colonies/μg DNA) |
|---|---|
| E. coli DH5α | Number of colonies (colonies/μg DNA) |
| E. coli BL21(DE3) | 1.5 x 105 |
[ Amplification of BioBrick parts : pSB1A2 and pSB1C3 ] 2010-09-05
1. Confirmed location : pSB1A2-BBa_E0040 (2010 Kit plate 1/ 14K) and pSB1C3-BBa_J04450 (2010 Kit plate 1/ 3A)
2. 20uL suspension by autoclaved distilled water
3. 3uL transformation to E. coli DH5α
4. Plating to LB(Amp100), LB(Cm25)
[ Genomic DNA extraction ] 2010-09-06
1. Inoculation for plasmid DNA purification
2. E. coli K12 genomic DNA extraction by AccuPrep® Genomic DNA Extraction Kit
3. Confirmation of genomic DNA by agarose gel electrophoresis (Figure 1)
4. Quantification of DNA concentration by NanoDrop : 137.5ng/μL
[ Plasmid DNA extraction : pSB1A3 and pSB1C3 ] 2010-09-07
1. Plasmid miniprep by LaboPass™ Plasmid Mini (Plasmid DNA purification kit)
2. Confirmation of extracted plasmids by agarose gel electrophoresis (Figure 2)
3. Quantification of DNA concentration by NanoDrop
[ PCR : promoters and reporter genes ] 2010-09-13 ~ 2010-09-16
1. PCR : PyodA-mAAA, PzntA-RFP(BBa_E1010) and ParsR-GFP(BBa_E0040)
| Reagent | Volume (μL) |
|---|---|
| 2.5mM dNTP | 3 |
| 10x buffer | 5 |
| Plasmid template (20ng/μL) | 2 |
| Primers (10pmole/μL) | 4 |
| α-Taq DNA polymerase (5U/μL) | 0.5 |
| D.W. | 35.5/total=50 |
| 95˚C(2’)-[95˚C(20”)-55˚C(20”)-72˚C(2’)]30-72˚C(5’)-4˚C |
2. Confirmation of PCR products by agarose gel electrophoresis (Figure 3)
3. Purified PCR products
4. Quantification of DNA concentration by NanoDrop
[ Digestion] 2010-09-17
1. Digestion of PCR products and pSB1A2
- 1) PyodA-mAAA : EcoRI and SpeI
- 2) PzntA-RFP : XbaI and PstI
- 3) pSB1A3 : EcoRI and PstI
| Reagent | Volume (μL) |
|---|---|
| DNA (about 30ng/μL) | 30 |
| 10x NEB buffer 2 | 5 |
| BSA (10mg/mL) | 0.5 |
| Appropriate 1st and 2nd restriction enzymes | 2 (each 1) |
| D.W. | 12.5 / total = 50 |
| Completely digestion at 37˚C for 2 hours (at least)
and stop at 80˚C for 20min |
2. Confirmation of digested products by agarose gel electrophoresis (Figure 4)
3. Quantification of DNA concentration by NanoDrop
[Chuseok, Korean thanksgiving day] 2010-09-20 ~ 2010-09
[ Ligation & Transformation ] 2010-09-27
1. Ligation of each parts : PyodA-mAAA, PzntA-RFP and pSB1A2
| Reagent | Volume (μL) |
|---|---|
| 10x T4 DNA ligase reaction buffer | 2 |
| T4 DNA ligase | 2 |
| Each of the digests | 2 + 2 + 2 = 8 |
| D.W. | 8 / total = 20 |
| Incubation at room temperature for 30min
and stop at 80˚C for 20min |
2. Transformation to E. coli DH5α
[ Confirmation of 1st cloning ] 2010-09-28
1. Check : the color of colonies (pSB1A2 : green, recombinant plasmid : white)
2. Inoculation of white colonies to 3mL LB(Amp100)
[ Plasmid DNA extraction : pSB1A2-( PyodA-mAAA-PzntA-RFP) ] 2010-09-29
1. Plasmid DNA purification by LaboPass™ Plasmid Mini
2. Confirmation of extracted plasmids by agarose gel electrophoresis (Figure 5)
3. Recombinant plasmid sequencing by COSMO GeneTech
[ Digestion ] 2010-10-01 ~ 2010-10-03
1. Check : recombinant plasmid sequence
2. Selection of correct clones
3. Digestion of PCR products(ParsR-GFP) and pSB1C3
- 1) ParsR-GFP : EcoRI and SpeI
- 2) pSB1C3 : EcoRI and PstI
| Reagent | Volume (μL) |
|---|---|
| DNA (about 30ng/μL) | 30 |
| 10x NEB buffer 2 | 5 |
| BSA (10mg/mL) | 0.5 |
| Appropriate 1st and 2nd restriction enzymes | 2 (each 1) |
| D.W. | 12.5 / total = 50 |
| Completely digestion at 37˚C for 2 hours (at least)
and stop at 80˚C for 20min |
4. Confirmation of digested products by agarose gel electrophoresis (Figure 6)
5. Quantification of DNA concentration by NanoDrop
6. Ligation of each parts : PyodA-mAAA-PzntA-RFP, ParsR-GFP and pSB1C3
| Reagent | Volume (μL) |
|---|---|
| 10x T4 DNA ligase reaction buffer | 2 |
| T4 DNA ligase | 2 |
| Each of the digests | 2 + 2 + 2 = 8 |
| D.W. | 8 / total = 20 |
| Incubation at room temperature for 30min
and stop at 80˚C for 20min |
7. Transformation to E. coli DH5α
[ Confirmation of 2nd cloning ] 2010-10-06
1. Check : the color of colonies (pSB1C3 : red, recombinant plasmid : white)
2. Inoculation of white colonies to 3mL LB(Amp100)
[ Plasmid DNA extraction : pSB1C3-( PyodA-mAAA-PzntA-RFP-ParsR-GFP) ] 2010-10-07
1. Plasmid DNA purification by LaboPass™ Plasmid Mini
2. Confirmation of extracted plasmids by agarose gel electrophoresis (Figure 7)
3. Recombinant plasmid full-sequencing by COSMO GeneTech
[ Completion : Heavy-metal detector ] 2010-10-18
1. Check : recombinant plasmid sequence
2. Selection of correct clones
3. Transformation to E. coli BL21(DE3) for expression test
 "
"