Template:UNIPV-Pavia/Project/results/Self-cleaving affinity tags to easily purify proteins
From 2010.igem.org
PHB production
Fusion protein synthesis
These experiments were performed to check bacterial growth and GFP synthesis rate of the following constructs (and efficiency of inducible system of some of them) in order to verify right protein folding.
Costitutive promoter devices
Methods
Inoculum (into 5 ml LB+Amp) from glycerol stock of:
- <partinfo>BBa_K300086</partinfo>
- <partinfo>BBa_K300088</partinfo>
- <partinfo>BBa_K300090</partinfo>
- <partinfo>BBa_K300099</partinfo>
- <partinfo>BBa_K173000</partinfo> (positive control)
- <partinfo>BBa_B0031</partinfo> (negative control)
They were let grow ON at +37°C, 220 rpm.
The following day cultures were diluted 1:100 and let grow again for about five hours at +37°C, 220 rpm.
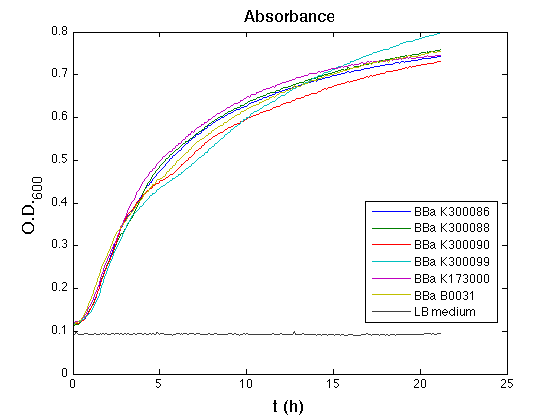
Optical density (O.D.) of each cell culture was than measured with TECAN Infinte F200. Samples were diluted in order to obtain the same O.D. equal to 0,02.
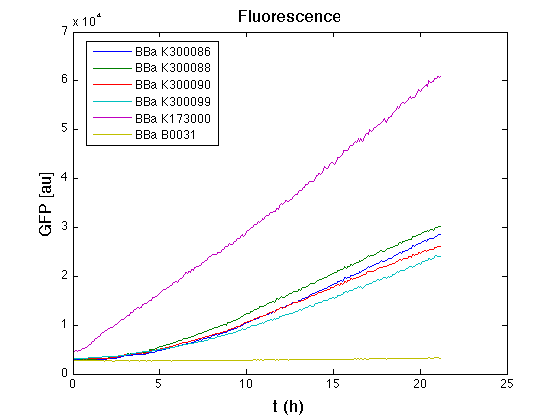
Than we performed a 21 hours' experiment with measurements of absorbance and green fluorescence every five minutes with TECAN Infinite F200. Each value shown is the mean of three measurements, from GFP data that of a non-fluorescent culture (negative control) was subtracted; cultures were shaken for 15 seconds every five minutes.
Results
| Culture | Doubling time [min.] |
|---|---|
| <partinfo>BBa_K173000</partinfo> | 77 |
| <partinfo>BBa_K300086</partinfo> | 74 |
| <partinfo>BBa_K300088</partinfo> | 75 |
| <partinfo>BBa_K300090</partinfo> | 76 |
| <partinfo>BBa_K300099</partinfo> | 76 |
| <partinfo>BBa_B0031</partinfo> | 69 |
Discussion
All cell cultures showed a similar growth curve and doubling time was computed as described here in order to have informations about the burden due to synthesis of such fusion proteins. It's possible to see that all doubling time are very similar; it's possible to assert that the expression of these BioBrick parts doesn't cause abnormal stress to the cells.
In GFP curve it's possible to appreciate that in <partinfo>BBa_K300086</partinfo>, <partinfo>BBa_K300088</partinfo>, <partinfo>BBa_K300090</partinfo>, <partinfo>BBa_K300099</partinfo> GFP accumulation it's very similar and it's significantly different from that of negative control <partinfo>BBa_B0031</partinfo>. These results show the right folding of the green fluorescent protein assembled downstream of the genetic circuit.
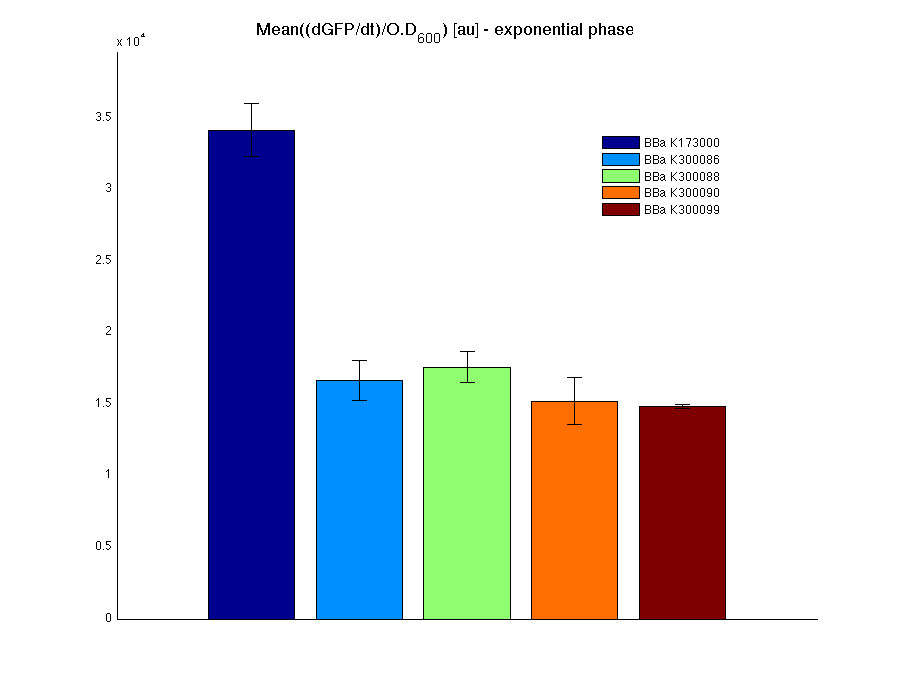
The mean protein synthesis rate was also computed over the growth exponential phase, showing again an appreciable GFP production rate that is about a half of the positive control.
HSL inducible devices
Methods
Inoculum (into 5 ml LB+Amp) from glycerol stock of:
- <partinfo>BBa_K300091</partinfo>
- <partinfo>BBa_K300092</partinfo>
- <partinfo>BBa_K173000</partinfo> (positive control)
- <partinfo>BBa_B0031</partinfo> (negative control)
They were let grow ON at +37°C, 220 rpm.
The following day cultures were diluted 1:100 and let grow again for about five hours at +37°C, 220 rpm.
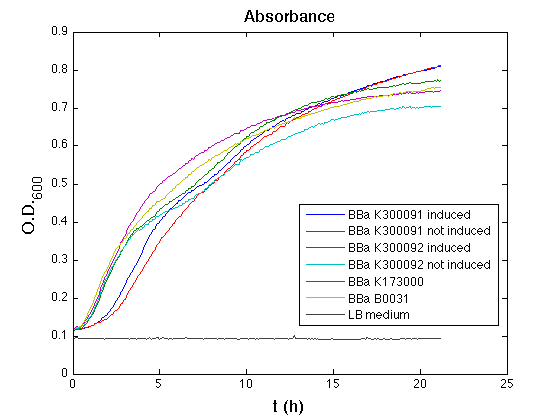
Optical density (O.D.) of each cell culture was than measured with TECAN Infinte F200. Samples were diluted in order to obtain the same O.D. equal to 0,02.
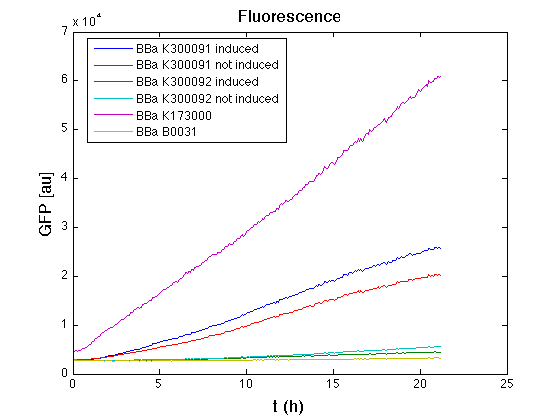
Than we performed a 21 hours' experiment with measurements of absorbance and green fluorescence every five minutes with TECAN Infinite F200. <partinfo>BBa_K300091</partinfo> and <partinfo>BBa_K300092</partinfo> circuits were induced 100nM with HSL directly into multiplate well. Each value shown is the mean of three measurements, from GFP data that of a non-fluorescent culture (negative control) was subtracted; cultures were shaken for 15 seconds every five minutes.
Results
| Culture | Doubling time [min.] |
|---|---|
| <partinfo>BBa_K173000</partinfo> | 75 |
| <partinfo>BBa_K300091</partinfo> induced | 121 |
| <partinfo>BBa_K300091</partinfo> not induced | 74 |
| <partinfo>BBa_K300092</partinfo> induced | 123 |
| <partinfo>BBa_K300092</partinfo> not induced | 72 |
| <partinfo>BBa_B0031</partinfo> | 74 |
Discussion
All cell cultures showed a similar growth curve and doubling time was computed as described here in order to have informations about the burden due to synthesis of such fusion proteins. It's possible to see that all doubling time are very similar except for induced cultures. In this case doubling time is much higher than posite control and not induced cultures; so it's possible to assert that in this case there's a kind of metabolic burden higher than in the others, maybe because of the inducible system.
In GFP curve it's possible to appreciate that in induced <partinfo>BBa_K300091</partinfo> and <partinfo>BBa_K300092</partinfo> GFP accumulation profile it's very similar and it's significantly different from that of negative control <partinfo>BBa_B0031</partinfo>. On the other hand not induced <partinfo>BBa_K300091</partinfo> and <partinfo>BBa_K300092</partinfo> show a profile very similar to the last one. These results show the right folding of the green fluorescent protein assembled downstream of the genetic circuit and that the inducible system works as expected.
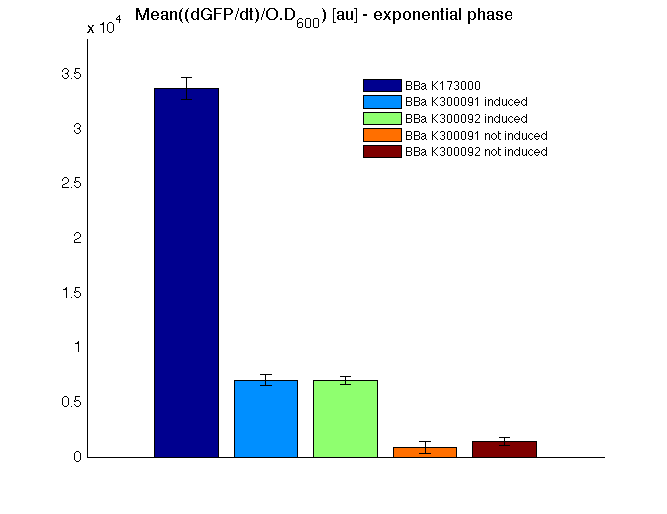
The mean protein synthesis rate was also computed over the growth exponential phase, showing again an GFP production rate that is different from negative control. Not induced <partinfo>BBa_K300091</partinfo> and <partinfo>BBa_K300092</partinfo> show a low GFP synthesis rate maybe due to 3OC6HSL inducible circuit leakage activity.
 "
"