|
JULY: WEEK 4
July, 19th
TECAN test showed that no RFP was produced from our parts, so all parts are potentially correct! For this reason we decided to sequence:
- I14-1 (Forward)
- I16-1 (Forward)
- I17-1 (Forward)
- I18-1 (Forward)
- I19-1 (Forward)
We also sequenced:
- I74C5-2 (Forward and Reverse)
- I84C5-2 (Forward and Reverse)
- I12-2 (Forward and Reverse)
These samples were prepared for sequencing (DNA was essicated) and sent to BMR genomics.
Trasformation of RING into:
- BW25141 (pir+)
- BW25142 (pir116)
- BW23474 (pir116)
- DH5alpha
- MG1655
Cultures were plated on:
- BW2514: Cm 34ug/ml
- BW25142: Cm 34ug/ml
- BW234741: Cm 34ug/ml
- DH5alpha: Cm 12,5ug/ml
- MG1655: Cm 12,5ug/ml
Inoculum of:
| I10-1 | I12-2 | I17-1
|
| I14-1 | <partinfo>BBa_J23110</partinfo> | I3-1
|
in 5ml LB+Amp. Cultures were grown ON 37°C 220 rpm.
July, 20th
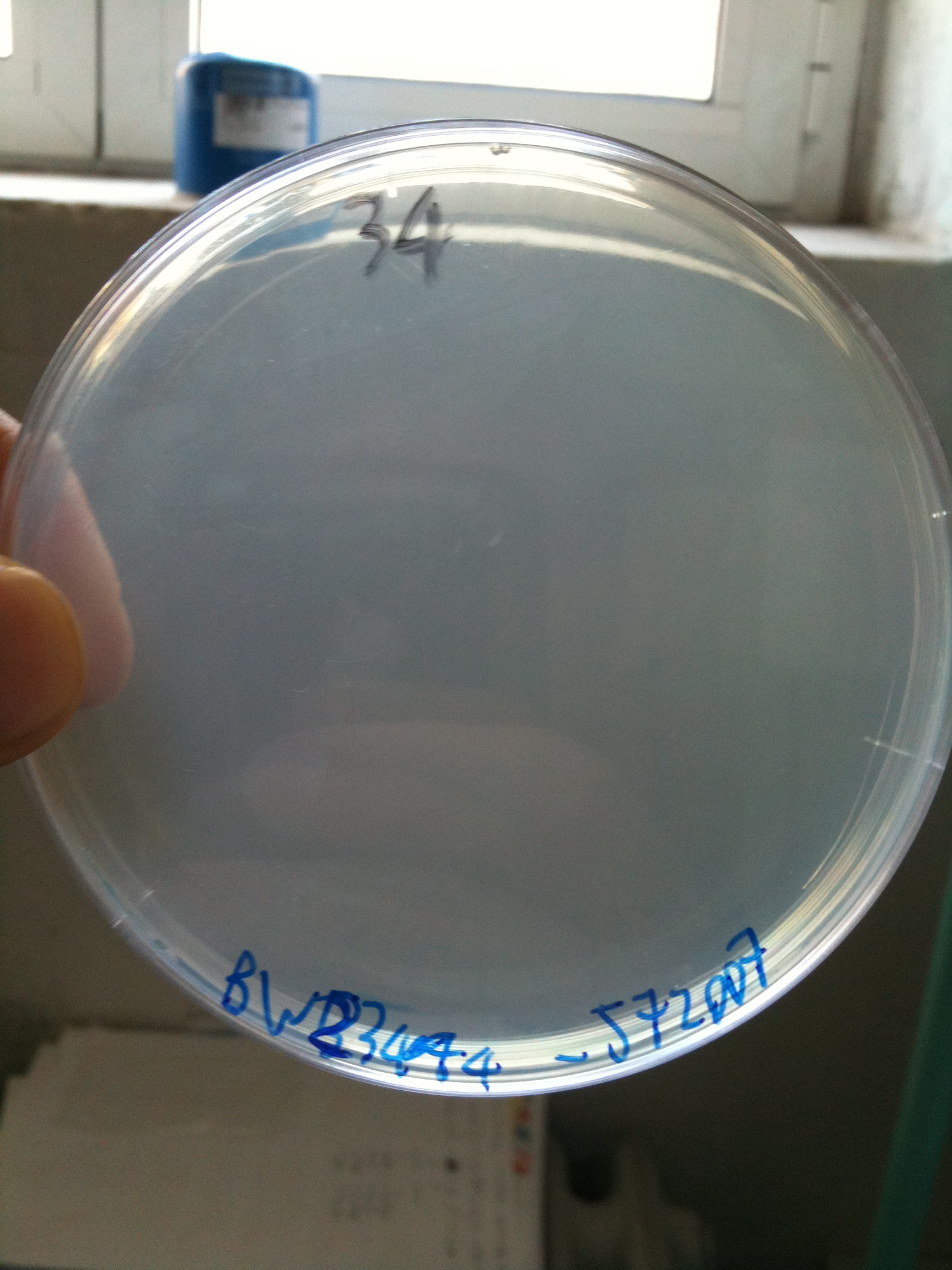 BW23474 transformed with <partinfo>BBa_J72007</partinfo> | 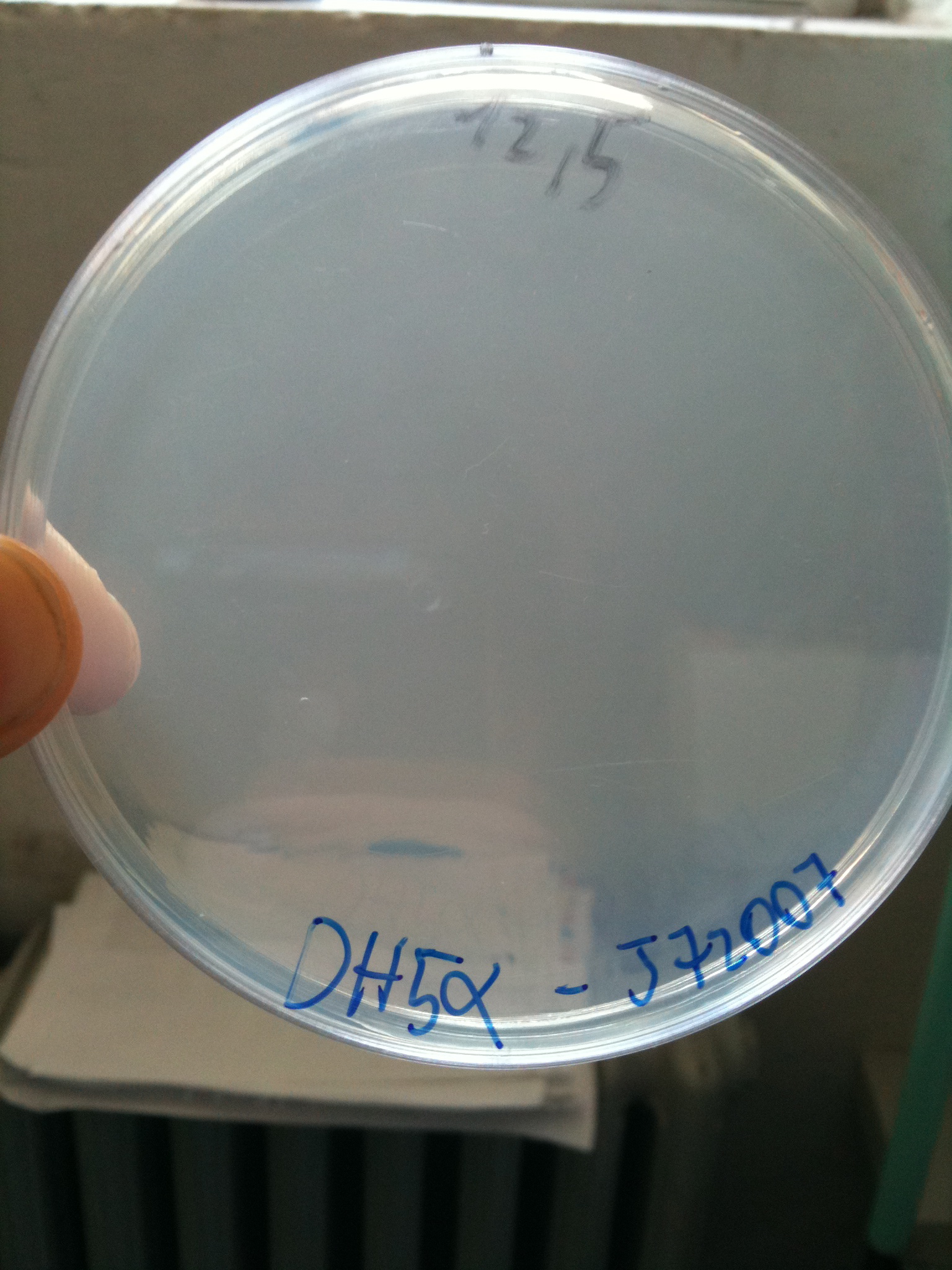 DH5alpha transformed with <partinfo>BBa_J72007</partinfo> |
Results for plates incubated ON, 37° C:
- BW23474 (pir116): showed colonies
- BW25141 (pir+): showed colonies
- BW25142 (pir116): showed colonies
- DH5alpha: didn't show colonies
- MG1655: didn't show colonies (even if there were a very few colonies that we suppose integrated the resistance of RING to survive - or the plate antibiotic wasn't homogeneous)
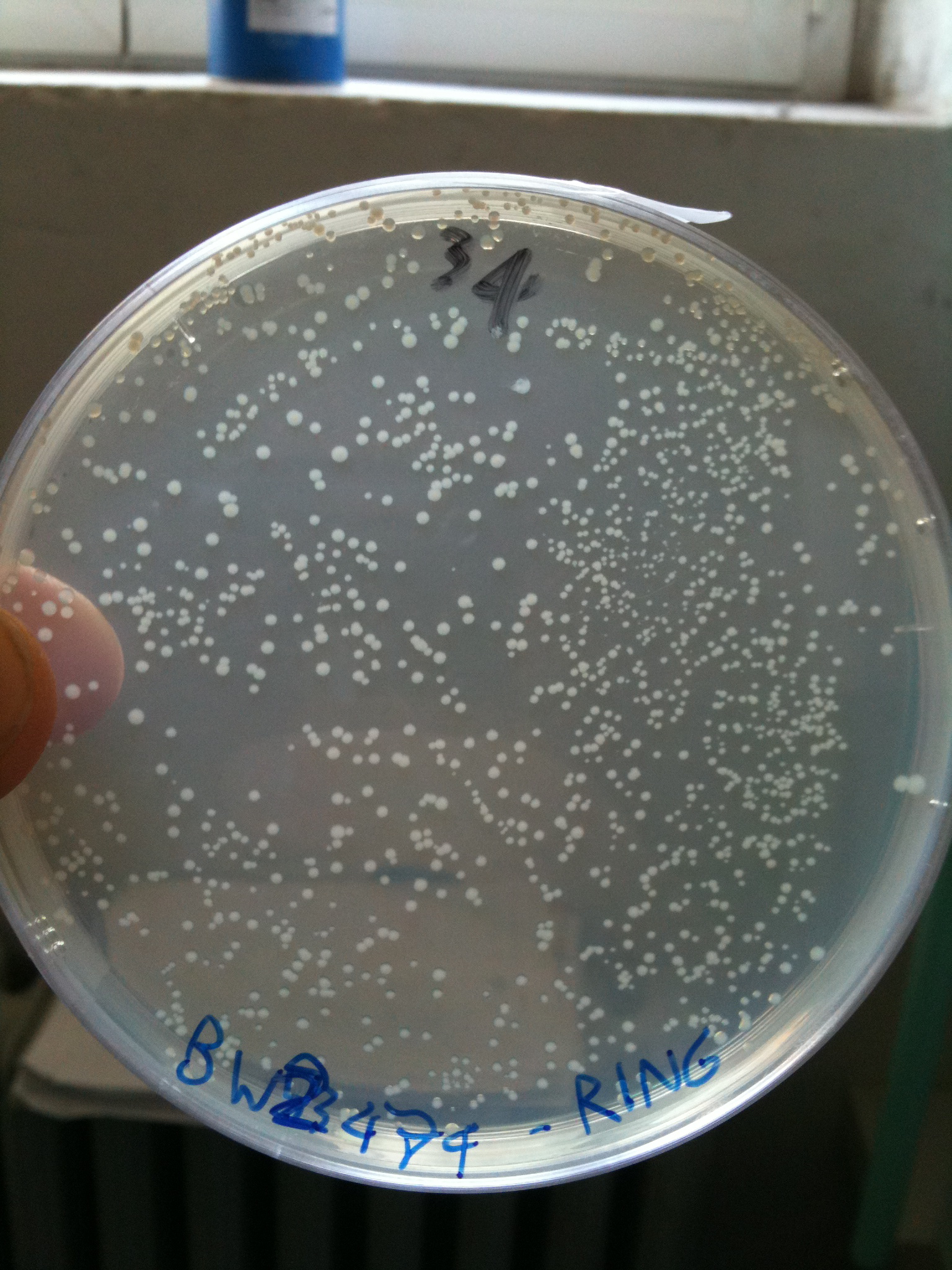 BW23474 transformed with RING | 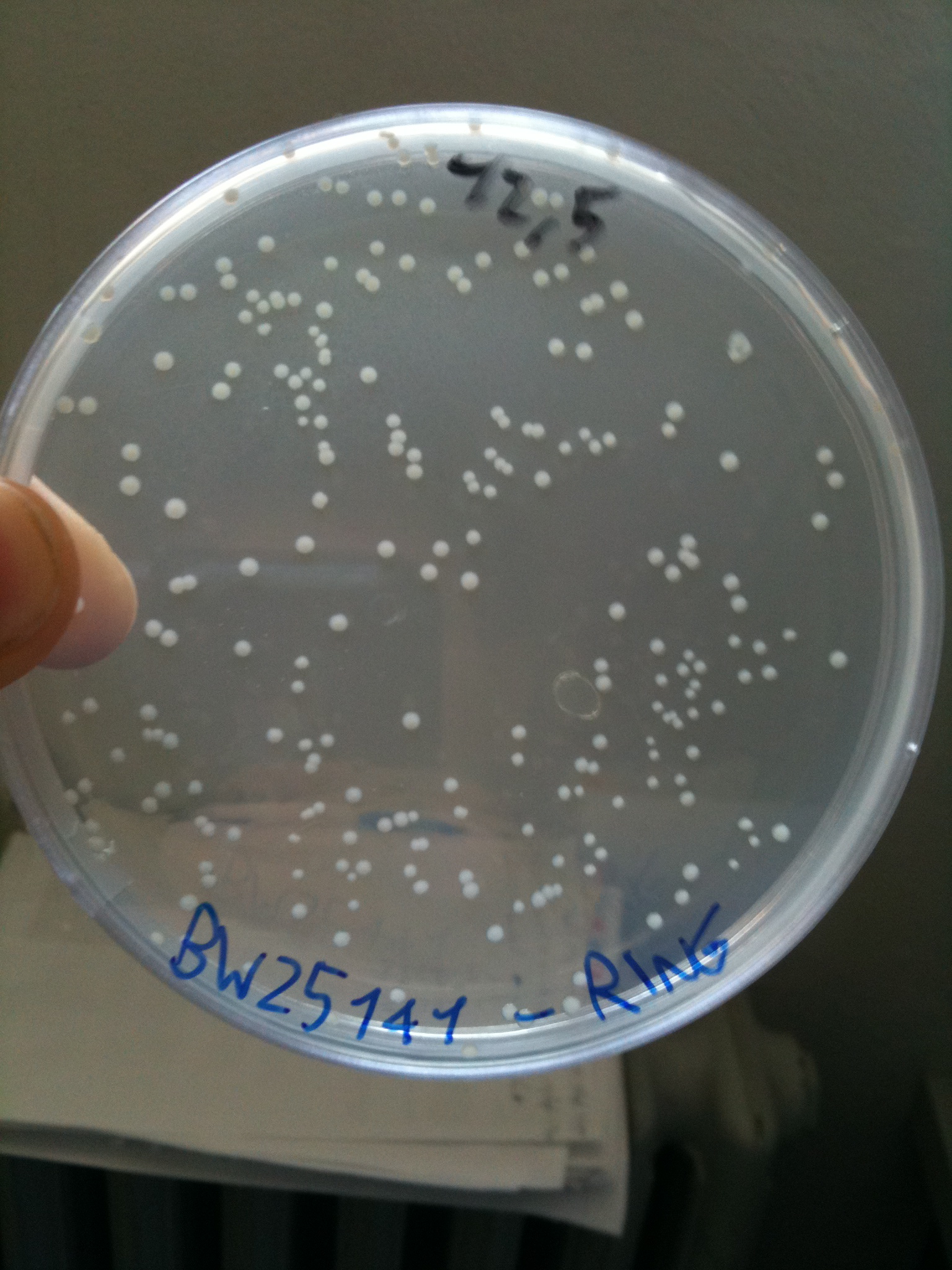 BW25141 transformed with RING | 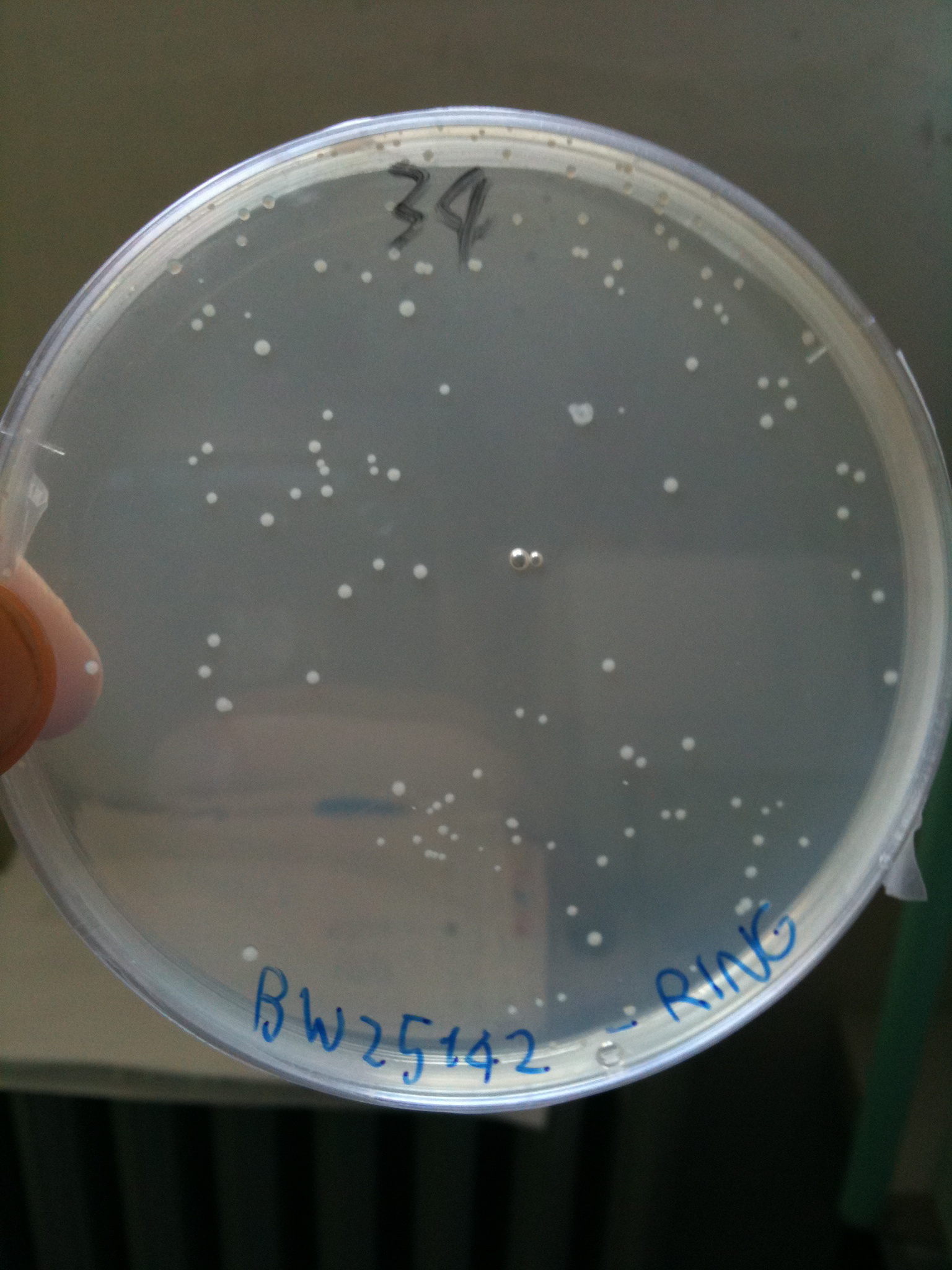 BW25142 transformed with RING |
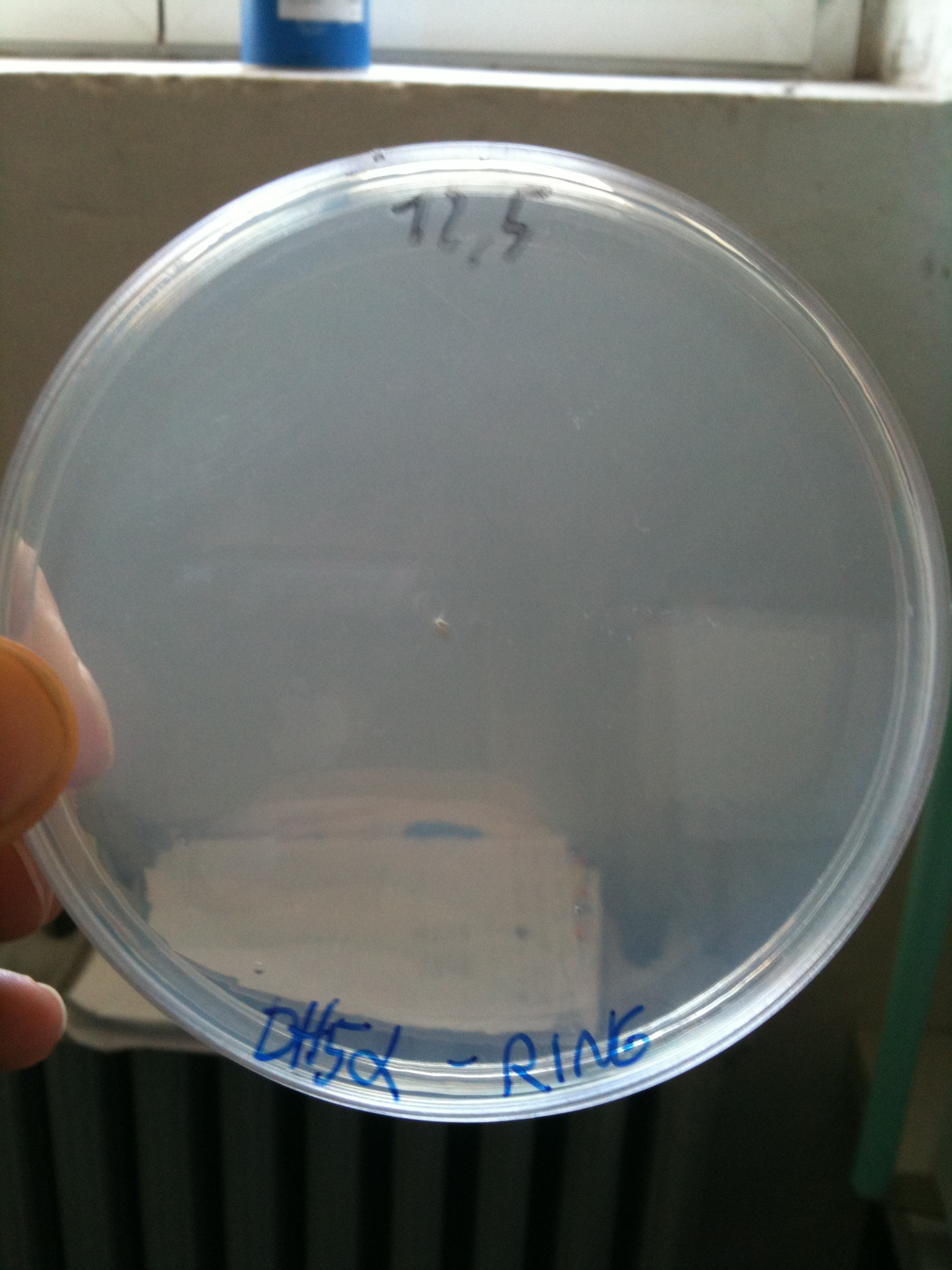 DH5alpha transformed with RING | 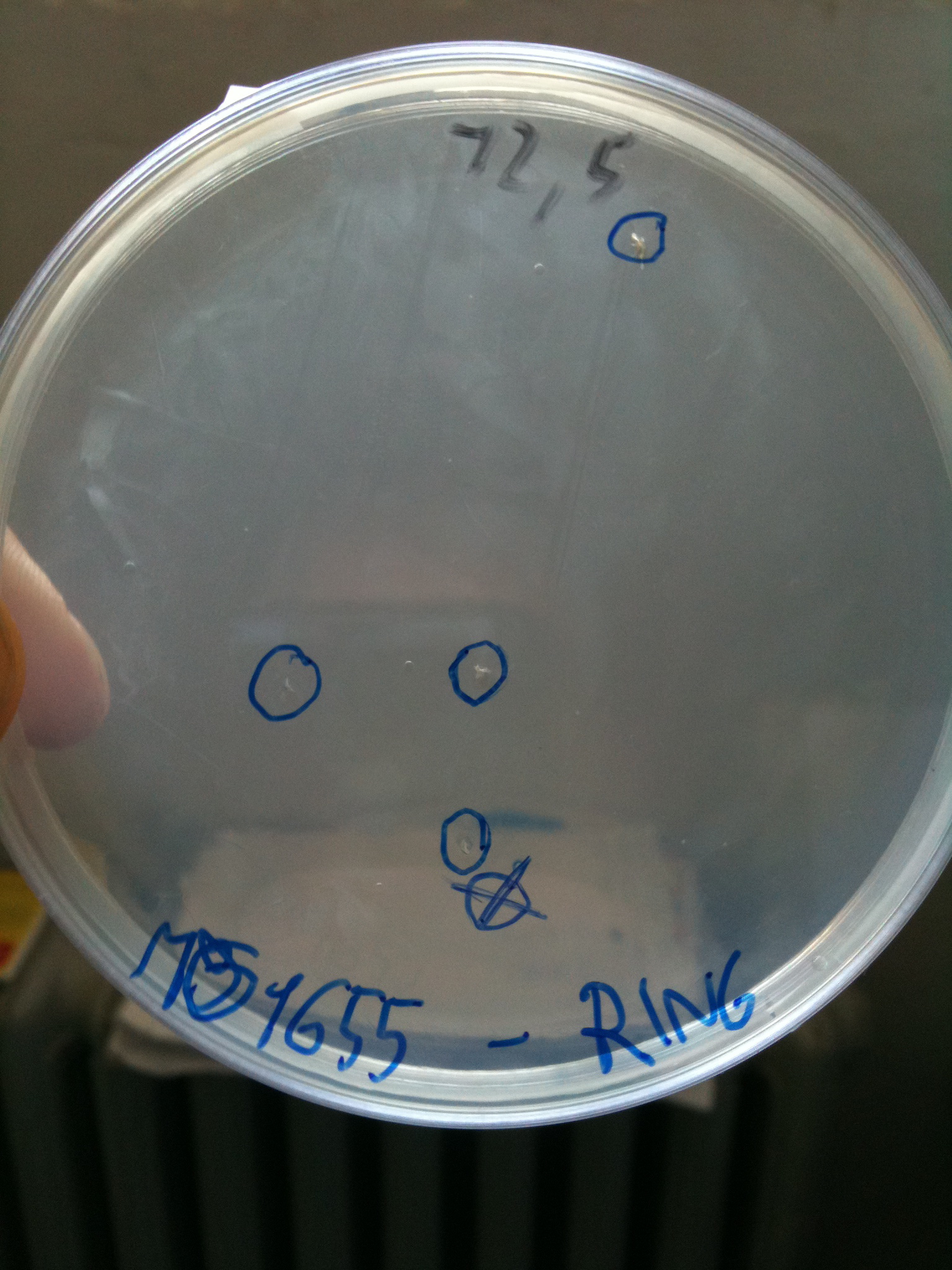 MG1655 transformed with RING |
Single colonies were picked from plates and grown in LB+Cm at proper concentration. MG1655 colonies were let grow in LB+Cm to check if they integrated the Cm resistance of RING.
MiniPrep was performed on cultures incubated yesterday, with following yields:
| Culture | Quantification
|
| I10-1 | 117.8 ng/ul
|
| I12-2 | 105,9 ng/ul
|
| I3-1 | 166,0 ng/ul
|
| I14-1 | 116,8 ng/ul
|
| I17-1 | 189,5 ng/ul
|
| <partinfo>BBa_J23110</partinfo> | 169,9 ng/ul
|
We retrieved from our freezer the following MiniPreps, with quantifications:
| Culture | Quantification
|
| I4C5 | 276 ng/ul
|
| I16-1 | 68,4 ng/ul
|
| I18-1 | 63,6 ng/ul
|
| I19-1 | 58,8 ng/ul
|
Digestion of:
| Culture | Kind | Final reaction volume (ul) | DNA (ul) | H20 (ul) | Enzyme 1 | Enzyme 2 | Buffer H
|
| <partinfo>BBa_J23110</partinfo> | Vector | 25 | 6 | 14,5 | 1 SpeI | 1 PstI | 2,5
|
| I3-1 | Vector | 25 | 10,5 | 10 | 1 XbaI | 1 PstI | 2,5
|
| 4C5 | Vector | 25 | 3,6 | 16,9 | 1 EcoRI | 1 PstI | 2,5
|
| I14-1 | Insert | 25 | 12,8 | 7,7 | 1 EcoRI | 1 PstI | 2,5
|
| I16-1 | Insert | 25 | 12,5 | 8 | 1 EcoRI | 1 PstI | 2,5
|
| I17-1 | Insert | 25 | 8 | 12,5 | 1 EcoRI | 1 PstI | 2,5
|
| I18-1 | Insert | 25 | 13 | 7,5 | 1 EcoRI | 1 PstI | 2,5
|
| I19-1 | Insert | 25 | 13 | 7,5 | 1 EcoRI | 1 PstI | 2,5
|
| I12-2 | Insert | 25 | 14 | 6,5 | 1 EcoRI | 1 PstI | 2,5
|
| I10-1 | Insert | 25 | 12,5 | 8 | 1 EcoRI | 1 PstI | 2,5
|
July, 21st
Tecan Test
July, 22nd
July, 23rd
July, 24th
Efficiency of transformation:
| Culture | Colonies
|
| BW25141 | 232 colonies
|
| BW25142 | 137 colonies
|
| BW23474 | 1436 colonies
|
July, 25th
|
|