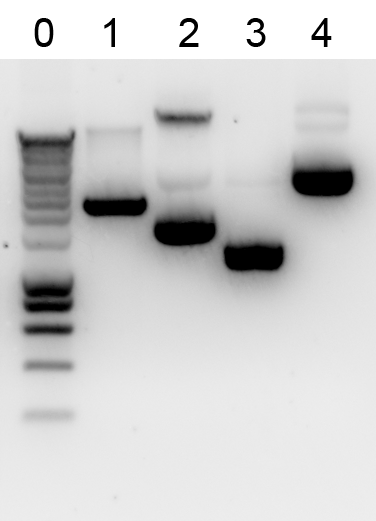Team:TU Delft/3 August 2010 content
From 2010.igem.org
(→Alkane degradation) |
|||
| Line 4: | Line 4: | ||
[[Image:TU_Delft_Pi2_Colony_PCR_retry_part_1.png|400px]] | [[Image:TU_Delft_Pi2_Colony_PCR_retry_part_1.png|400px]] | ||
| + | |||
| + | * BBa_K398305 = Alks -> E0240 | ||
| + | * BBa_K398101 = bbc1 -> J61101 | ||
| + | * BBa_K398402 = PhPFDalpha -> J61101 | ||
| + | |||
| + | Lane Description: | ||
| + | {|style="color:black; background-color:white;" cellpadding="5" cellspacing="0" border="1" | ||
| + | |'''#''' | ||
| + | |'''Description''' | ||
| + | |'''Expected Length (bp)''' | ||
| + | |'''Primers''' | ||
| + | |'''Status''' | ||
| + | |'''Remarks''' | ||
| + | |- | ||
| + | |M | ||
| + | |SmartLadder | ||
| + | |n/a | ||
| + | |n/a | ||
| + | |n/a | ||
| + | | | ||
| + | |- | ||
| + | |1-8 | ||
| + | |BBa_K398305 | ||
| + | |3772 | ||
| + | |G00101 + G00100 | ||
| + | | | ||
| + | | | ||
| + | |- | ||
| + | |M | ||
| + | |SmartLadder | ||
| + | |n/a | ||
| + | |n/a | ||
| + | |n/a | ||
| + | | | ||
| + | |- | ||
| + | |9-16 | ||
| + | |BBa_K398101 | ||
| + | |1007 | ||
| + | |G00101 + G00100 | ||
| + | | | ||
| + | | | ||
| + | |- | ||
| + | |M | ||
| + | |SmartLadder | ||
| + | |n/a | ||
| + | |n/a | ||
| + | |n/a | ||
| + | | | ||
| + | |- | ||
| + | |17-24 | ||
| + | |BBa_K398101 | ||
| + | |1007 | ||
| + | |G00101 + G00100 | ||
| + | | | ||
| + | | | ||
| + | |} | ||
| + | |||
| + | [[Image:TU_Delft_Pi2_Colony_PCR_retry_part_2.png|400px]] | ||
| + | |||
| + | * BBa_K398403 = PhPFDbeta -> J61101 | ||
=Alkane degradation= | =Alkane degradation= | ||
Revision as of 12:30, 4 August 2010
Alkane Sensing, Solvent Tolerance and Salt Tolerance
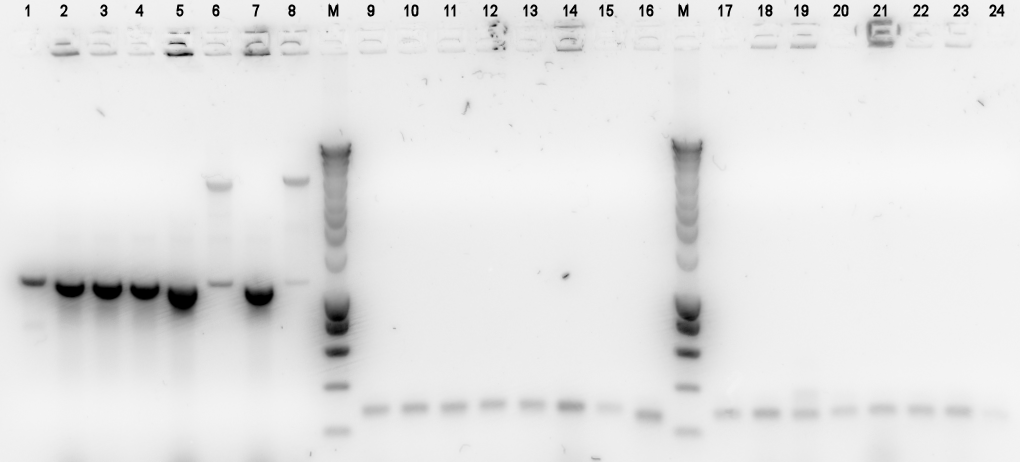
by Pieter Because yesterday's Colony PCRs all turned out negative, we decided to pick an additional 8 colonies per plate and perform Colony PCR on them.
- BBa_K398305 = Alks -> E0240
- BBa_K398101 = bbc1 -> J61101
- BBa_K398402 = PhPFDalpha -> J61101
Lane Description:
| # | Description | Expected Length (bp) | Primers | Status | Remarks |
| M | SmartLadder | n/a | n/a | n/a | |
| 1-8 | BBa_K398305 | 3772 | G00101 + G00100 | ||
| M | SmartLadder | n/a | n/a | n/a | |
| 9-16 | BBa_K398101 | 1007 | G00101 + G00100 | ||
| M | SmartLadder | n/a | n/a | n/a | |
| 17-24 | BBa_K398101 | 1007 | G00101 + G00100 |
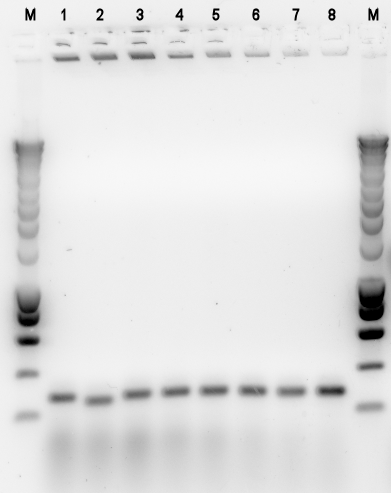
- BBa_K398403 = PhPFDbeta -> J61101
Alkane degradation
Yesterday's digestions showed that BioBrick 007 was not the expected length. From this it was concluded that the plasmid thus does not contain the desired insert, so we will make the BioBrick again. This was done, starting with a digestion. An extra cut was done in the backbone of alkB2 to decrease the chances of religation.
| # | Sample | Enzyme 1 | Enzyme 2 | Enzyme 3 | Buffer | BSA | Needed fragment |
| 1 | J61100 | SpeI | PstI | ✗ | 2 (Biolabs) | ✓ | ‘S – pSB1A2 - J61100– P’ |
| 2 | 1 μg alkB2 | XbaI | PstI | AseI | 2 (Biolabs) | ✓ | ‘X – alkB2 – P’ |
The digestions were done during 2 hours, and checked on a gel:
Lane description
| # | Description | Expected Length (bp) | Status |
| 0 | Smartladder (5μl) | ||
| 1 | ‘S – pSB1A2 - J61100 – P’ | 2116 | ✓ |
| 2 | undigested pSB1A2-J61100 | ✓ | |
| 3 | ‘X – alkB2 – P’ | 1263 | ✓ |
| 4 | Undigested alkB2 | ✓ |
Directly following the digestion, the products were ligated for 4 hours:
| # | BioBrick | Fragment 1 | Fragment 2 |
| 1 | pSB1A2-J61100-alkB2 | ‘X – alkB2 – P’ | ‘S – pSB1A2-J61100 – P’ |
10 μL of this ligation mix aswell as yesterday's ligations were used to transform Top10 competent cells.
 "
"