Team:TU Delft/15 July 2010 content
From 2010.igem.org
(→Characterization of Anderson RBS sequences) |
(→Characterization of Anderson RBS sequences) |
||
| Line 95: | Line 95: | ||
<h5>Fluorescence measurements attempt #2:</h5> | <h5>Fluorescence measurements attempt #2:</h5> | ||
| - | 1. [https://2010.igem.org/Team:TU_Delft#/blog?blog=14_July_2010 Last night's] cultures of TOP10 carrying K398500, K398501, K398502, K398503, K398504 and I13401 were measured for biomass (OD600) and diluted to an OD600 of 0.1. | + | 1. [https://2010.igem.org/Team:TU_Delft#/blog?blog=14_July_2010 Last night's] cultures of TOP10 carrying K398500, K398501, K398502, K398503, K398504 and I13401 were measured for biomass (OD600) and diluted to an OD600 of about 0.1 (see exact OD600 measurement of dilutions in table below). |
| - | 2. Aliquots of 175 uL of the dilutions were pipetted into the wells of a 96-wells plate in three-fold. | + | {| style="color:black; background-color:white;" cellpadding="5" cellspacing="0" border="1" |
| + | |'''#''' | ||
| + | |'''OD600''' | ||
| + | |- | ||
| + | |K398500 | ||
| + | |0.102 | ||
| + | |- | ||
| + | |K398501 | ||
| + | |0.108 | ||
| + | |- | ||
| + | |K398502 | ||
| + | |0.108 | ||
| + | |- | ||
| + | |K398503 | ||
| + | |0.107 | ||
| + | |- | ||
| + | |K398504 | ||
| + | |0.098 | ||
| + | |- | ||
| + | |I13401 | ||
| + | |0.109 | ||
| + | |} | ||
| + | |||
| + | 2. Aliquots of 175 uL of the dilutions were pipetted into the wells of a 96-wells plate in three-fold. The plate was covered with a transparent film to prevent evaporation. | ||
3. The fluorescence and OD600 were measured for 16.30 hours with intervals 10 minutes by means of the Biotek Synergy plate reader with the excitation filter set at 485nm and the emission filter at 520nm. | 3. The fluorescence and OD600 were measured for 16.30 hours with intervals 10 minutes by means of the Biotek Synergy plate reader with the excitation filter set at 485nm and the emission filter at 520nm. | ||
| Line 103: | Line 126: | ||
<h5>Assembly of reference construct:</h5> | <h5>Assembly of reference construct:</h5> | ||
K081005 yielded no transformants and was thus transformed a second time. | K081005 yielded no transformants and was thus transformed a second time. | ||
| - | Transformants of J23100 were single colony PCRed (see gel image) and used as inoculate for 5 mL of LB for later plasmid isolation and [[Team:TU_Delft/protocols/freezing_bacterial_stocks|-80 °C stocks]]. | + | Transformants of J23100 were single colony PCRed (see gel image below) and used as inoculate for 5 mL of LB for later plasmid isolation and [[Team:TU_Delft/protocols/freezing_bacterial_stocks|-80 °C stocks]]. |
The PCR amplification product of K081005 was digested using EcoRI and SpeI. | The PCR amplification product of K081005 was digested using EcoRI and SpeI. | ||
| Line 109: | Line 132: | ||
<h5>Positive control</h5> | <h5>Positive control</h5> | ||
| - | Transformants of I13522 were single colony PCRed (see gel image) and used as inoculate for 5 mL of LB for later plasmid isolation and [[Team:TU_Delft/protocols/freezing_bacterial_stocks|-80 °C stocks]]. | + | Transformants of I13522 were single colony PCRed (see gel image below) and used as inoculate for 5 mL of LB for later plasmid isolation and [[Team:TU_Delft/protocols/freezing_bacterial_stocks|-80 °C stocks]]. |
<h5>Images of the day</h5> | <h5>Images of the day</h5> | ||
Revision as of 20:02, 20 July 2010
Contents |
Lab work
Ordered DNA + Solvent Tolerance and Hydrocarbon Sensing
Some of transformations of yesterday containing the different ligations gave colonies (1 a 2 per plate). We picked as many colonies as possible of every plate and performed a colony PCR overnight to check which colonies contained the right insert. At the same time we grow them overnight in 5 mL LB medium containing the appropriate antibiotic.
Emulsifier
The results from the Colony PCR were not conclusive yesterday. That is why we decided to isolate the plasmid using Birnboim protocol. We used 3 mL of the bacterial cells to make -80 °C stocks.
The following plasmid concentrations were obtained:
| BioBrick | Composed of | Concentration (ng/μL) |
| K398203 | OprG-B0015 (transformant 6) | |
| K398203 | OprG-B0015 (transformant 7) | |
| K398204 | AlnA-B0015 (transformant 11) |
The plasmids were subsequently digested with an enzyme that characteristic digest for all different plasmids. The plasmids were digested with HindIII. This should yield 3 bands in the lane with the plasmid pSB1T3 containing J04450 (neg control), 1 band in the lane with AlnA-B0015 and 2 bands in the lane with OprG-B0015 on gel.
| # | Digestion reaction | Used Buffer |
| 1 | 1.5 μg pSB1T3 | Buffer 2 + BSA (BioLabs) |
| 2 | 1 μg OprG-B0015 (transformant 6) | Buffer 2 + BSA (BioLabs) |
| 3 | 1 μg OprG-B0015 (transformant 7) | Buffer 2 + BSA (BioLabs) |
| 4 | 1 μg AlnA-B0015 (transformant 11) | Buffer 2 + BSA (BioLabs) |
1% Agarose gel of digestion products:
Lane description:
| # | Description | Expected Length (bp) |
| 1 | SmarLadder marker (5 μL) | |
| 2 | Undigested pSB1T3 (5 μL + 1 μL loadingbuffer) | |
| 3 | Digested pSB1T3 (10 μL + 1 μL loadingbuffer) | 1972, 1176 |
| 4 | Undigested OprG-B0015 (transformant 6) (5 μL + 1 μL loadingbuffer) | |
| 5 | Digested OprG-B0015 (transformant 6) (10 μL + 2 μL loadingbuffer) | 1828, 1475, 222 |
| 6 | Digested OprG-B0015 (transformant 7) (10 μL + 2 μL loadingbuffer) | 1828, 1475, 222 |
| 7 | Undigested AlnA-B0015 (transformant 11) (5 μL + 1 μL loadingbuffer) | |
| 8 | Digested AlnA-B0015 (transformant 11) (10 μL + 2 μL loadingbuffer) | 3511 |
Characterization of Anderson RBS sequences
Fluorescence measurements attempt #2:
1. Last night's cultures of TOP10 carrying K398500, K398501, K398502, K398503, K398504 and I13401 were measured for biomass (OD600) and diluted to an OD600 of about 0.1 (see exact OD600 measurement of dilutions in table below).
| # | OD600 |
| K398500 | 0.102 |
| K398501 | 0.108 |
| K398502 | 0.108 |
| K398503 | 0.107 |
| K398504 | 0.098 |
| I13401 | 0.109 |
2. Aliquots of 175 uL of the dilutions were pipetted into the wells of a 96-wells plate in three-fold. The plate was covered with a transparent film to prevent evaporation.
3. The fluorescence and OD600 were measured for 16.30 hours with intervals 10 minutes by means of the Biotek Synergy plate reader with the excitation filter set at 485nm and the emission filter at 520nm.
Assembly of reference construct:
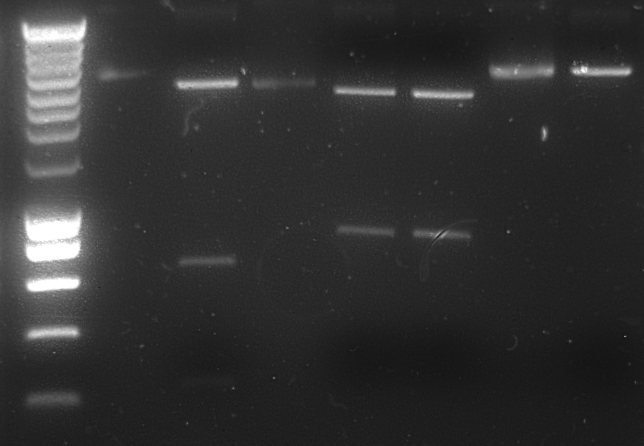
K081005 yielded no transformants and was thus transformed a second time. Transformants of J23100 were single colony PCRed (see gel image below) and used as inoculate for 5 mL of LB for later plasmid isolation and -80 °C stocks.
The PCR amplification product of K081005 was digested using EcoRI and SpeI. 2 ug of I13401 in pSB1A2 was digested using EcoRI and XbaI.
Positive control
Transformants of I13522 were single colony PCRed (see gel image below) and used as inoculate for 5 mL of LB for later plasmid isolation and -80 °C stocks.
Images of the day
1% Agarose gel of scPCR product:
Lane descriptions:
| # | Description | Expected Length (bp) |
| 1 | SmartLadder marker (5 μL) | |
| 2 | scPCR product of J23100 transformant #1 (5 μL + 1 μL loadingbuffer) | |
| 3 | scPCR product of J23100 transformant #2 (5 μL + 1 μL loadingbuffer) | |
| 4 | scPCR product of I13522 transformant #1 (5 μL + 1 μL loadingbuffer) | |
| 5 | EZ Load Ladder (5 μL) |
From the gel it can be concluded that the transformants carry the desired plasmids with the proper BioBrink inserts.
 "
"