Team:Stockholm/Project Idea/Proteins
From 2010.igem.org
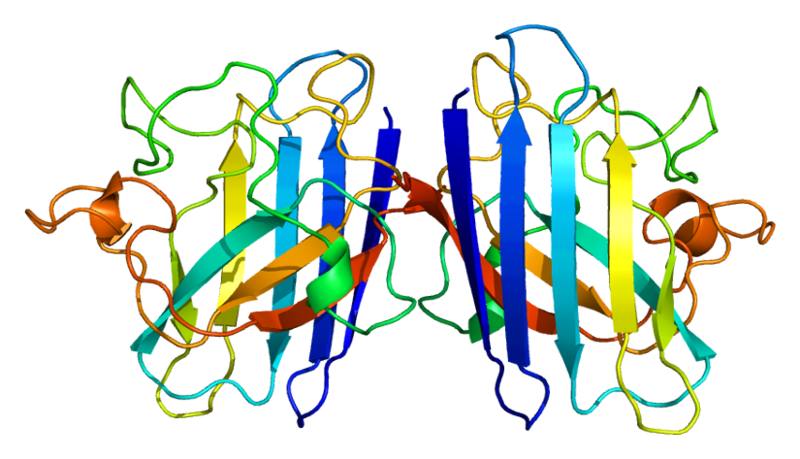
ProteinsSuperoxide dismutase 1 (SOD1)Human soluble Superoxide dismutase 1 (SOD1) is a soluble cytoplasmic protein functional as a homodimer that binds copper and zink ions. SOD1 catalyzes the reaction O-2 + O-2 + 2H+ → H2O2 + O2, protecting the cell from oxidative damage. SOD1 was first cloned and expressed in Escherichia coli by Hallewell et al., (1985).
Yeast copper chaperon (yCCS)Yeast copper chaperon protein (yCCS) is a helper chaperon specific for copper/zinc superoxide dismutase located to the cytoplasm. yCCS generates fully metallized, active SOD1 proteins that in turn protects the cell from oxidative damage. yCCS has been shown to successfully mediate the delivery of copper ions to human SOD1 (Ahl et al. 2003). Co-expression of SOD1 and yCCS yields proteins with higher copper contents, leading to increased activity and more stable proteins.
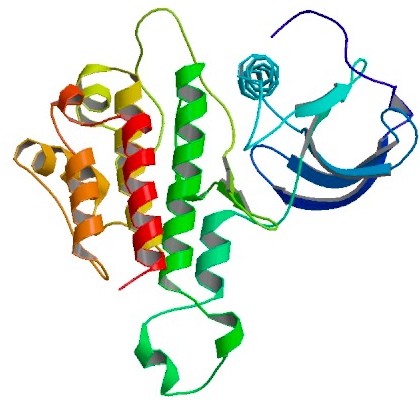
Human basic fibroblast growth factor (bFGF)
Protein A, z domain
[[Image:|300px|thumb|right|]]
First reported by:
IgG protease, IdeS
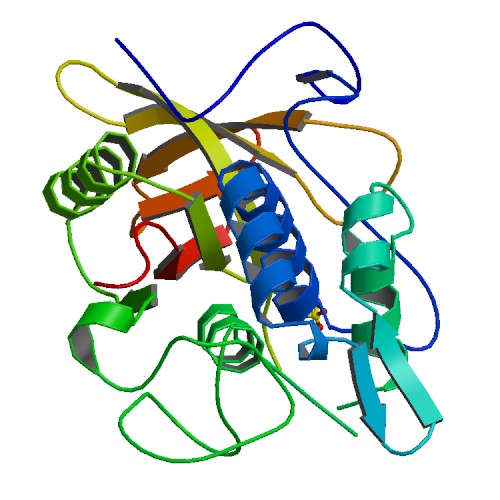 3D structure of IdeS. Primary citation Wenig et al. 2004
First reported by:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 |

|
 |

|
 |

|

|

|
 "
"