Team:Stockholm/Project Idea/Proteins
From 2010.igem.org
m (→Human basic fibroblast growth factor (bFGF)) |
m (→IgG protease (IdeS)) |
||
| (2 intermediate revisions not shown) | |||
| Line 116: | Line 116: | ||
|55,439 Da (full protein) | |55,439 Da (full protein) | ||
|- | |- | ||
| - | |'''Fasta''' | + | |'''Fasta'''<br /><br><br><br><br><br> |
| - | |[http://www.uniprot.org/uniprot/P38507.fasta Protein A] (full protein)<br /> | + | |[http://www.uniprot.org/uniprot/P38507.fasta Protein A] (full protein)<br /><br><br><br><br><br> |
| - | + | ||
| - | + | ||
|} | |} | ||
| Line 145: | Line 143: | ||
|37,977 Da | |37,977 Da | ||
|- | |- | ||
| - | |'''Fasta''' | + | |'''Fasta'''<br /><br><br><br><br><br> |
| - | |[http://www.ncbi.nlm.nih.gov/protein/209559219?report=fasta IdeS]<br /> | + | |[http://www.ncbi.nlm.nih.gov/protein/209559219?report=fasta IdeS]<br /><br><br><br><br><br> |
| - | + | ||
| - | + | ||
|} | |} | ||
Latest revision as of 01:19, 28 October 2010
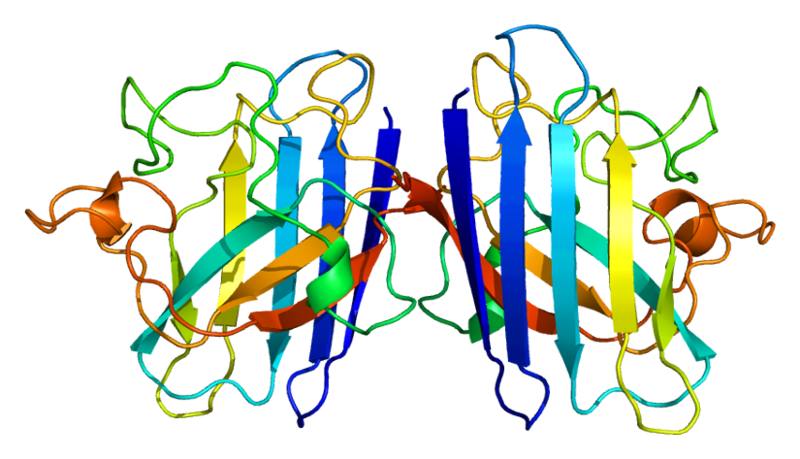
ProteinsSuperoxide dismutase 1 (SOD1)Human soluble Superoxide dismutase 1 (SOD1) is a soluble cytoplasmic protein functional as a homodimer that binds copper and zink ions. SOD1 catalyzes the reaction O-2 + O-2 + 2H+ → H2O2 + O2, protecting the cell from oxidative damage. SOD1 was first cloned and expressed in Escherichia coli by Hallewell et al., (1985).
Yeast copper chaperon (yCCS)Yeast copper chaperon protein (yCCS) is a helper chaperon specific for copper/zinc superoxide dismutase located to the cytoplasm. yCCS generates fully metallized, active SOD1 proteins that in turn protects the cell from oxidative damage. yCCS has been shown to successfully mediate the delivery of copper ions to human SOD1 (Ahl et al. 2003). Co-expression of SOD1 and yCCS yields proteins with higher copper contents, leading to increased activity and more stable proteins.
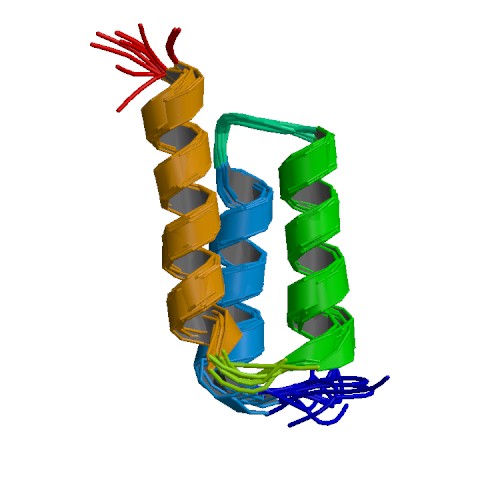
Human basic fibroblast growth factor (bFGF)
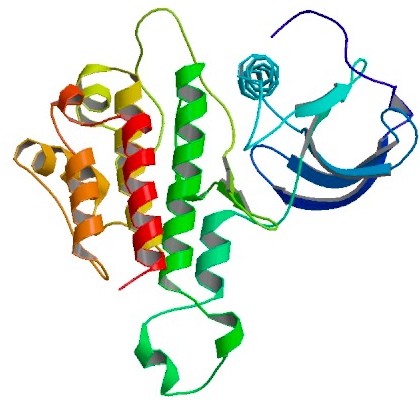
Protein A, z domain
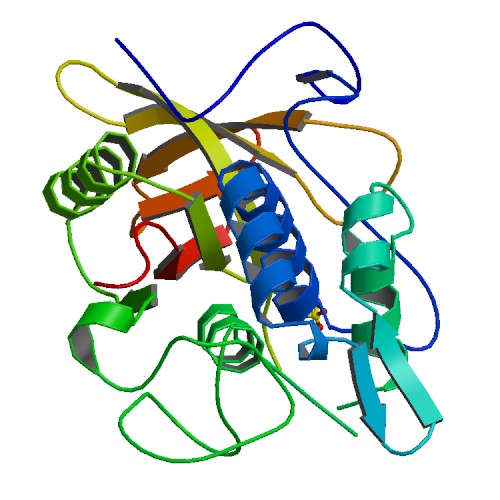
IgG protease (IdeS)
Cell penetrating peptidesThese cell-penetrating peptides, (CPPs) may be used in N- and C-terminal fusions with full-length proteins to create transduction proteins with the ability to permeate the lipid bilayer of various cell types, making them potential gene or protein delivery vectors.
TAT cell penetrating peptide (TAT)Purified full-length TAT fusion proteins expressed in Escherichia coli have been shown to successfully translocate into several human cell types, including all cells found in whole blood, as well as bone marrow stem cells and osteoblasts, while still retaining the fused protein's activity (Nagahara et al. 1998). The mechanism for transduction over the bilipid membrane is still a matter of debate, but has been suggested to occur through macropinocytosis, a specialized form of endocytosis (Gump and Dowdy, 2007). TAT is an 11-amino acid derivative from the Human Immunodeficiency Virus 1 (HIV-1) trans-activating transcriptional activator (Tat) (Green and Loewenstein, 1988; Nagahara et al. 1998). This part was back translated from the corresponding amino acid sequence and optimized for expression in Escherichia coli. Codon usage has been varied for repetitive amino acids to enable DNA synthesis.
Low molecular weight protamine (LMWP)Enzymatically prepared LMWP chemically conjugated to ovalbumin (OVA) and bovine serum albumin (BSA) have previously been shown to penetrate the lipid bilayer of human keratinocytes, as well as to successfully permeate mouse skin epidermis (Huang et al., 2010). Furthermore, LMWP/pDNA complexes can efficiently penetrate into human embryonic kidney cells (Park et al., 2003). As LMWP has been shown to be neither toxic nor immunogenic (Chang et al. a, 2001; Chang et al. b, 2001; Lee et al., 2001), it may be used as a potential vaccine, drug or gene delivery vector. LMWP is a 14-amino acid derivative from Rainbow trout (Oncorhynchus mykiss) protamine, an arginine-rich protein that replaces histones in chromatin during spermatogenesis (McKay et al., 1986; Byun et al., 1999). This part was back translated from the corresponding amino acid sequence and optimized for expression in Escherichia coli. Codon usage has been varied for repetitive amino acids to enable DNA synthesis.
Transportan 10 (Tp10)Chemically synthesized Tp10 peptides conjugated to different cargo, including pDNA and protein, have been shown to efficiently penetrate the lipid bilayer of both human and mouse cells (Kilk et al., 2005). Membrane permeation is both energy and temperature independent (Hällbrink et al., 2001). The exact mechanism for penetration is still unclear (Yandek et al., 2007). Tp10 is a 21-amino acid derivative from the parent peptide transportan (originally known as galparan), which is a peptide chimera of the neuropeptide galanin and the wasp venom peptide mastoparan (Soomets et al., 2000; Langel et al., 1996). This part was back translated from the corresponding amino acid sequence and optimized for expression in Escherichia coli. Codon usage has been varied for repetitive amino acids to enable DNA synthesis.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 |

|
 |

|
 |

|

|

|
 "
"