Team:Michigan/Oil Sands
From 2010.igem.org
| Line 39: | Line 39: | ||
| - | | - | ||
| - | | - | ||
| - | |||
!Week 4 | !Week 4 | ||
| - | | - | ||
| Line 48: | Line 47: | ||
| - | | - | ||
| - | | - | ||
| - | |||
!Week 5 | !Week 5 | ||
|[[Team:Michigan/Oil_Sands#7/12/2010|7/25/2010]] | |[[Team:Michigan/Oil_Sands#7/12/2010|7/25/2010]] | ||
| Line 57: | Line 55: | ||
|[[Team:Michigan/Oil_Sands#7/14/2010|7/30/2010]] | |[[Team:Michigan/Oil_Sands#7/14/2010|7/30/2010]] | ||
| - | | - | ||
| - | |||
|} | |} | ||
==Oil Sands Lab Notebook== | ==Oil Sands Lab Notebook== | ||
Revision as of 00:10, 31 July 2010
| Sunday | Monday | Tuesday | Wednesday | Thursday | Friday | Saturday | |||||||||||||||||
| Week 1 | - | 6/28/2010 | 6/29/2010 | 6/30/2010 | 7/1/2010 | 7/2/2010 | - | ||||||||||||||||
| Week 2 | - | - | - | 7/7/2010 | 7/8/2010 | 7/9/2010 | 7/10/2010 | ||||||||||||||||
| Week 3 | - | 7/12/2010 | 7/13/2010 | 7/14/2010 | - | - | - | Week 4 | - | - | - | - | - | - | - | Week 5 | 7/25/2010 | 7/26/2010 | 7/27/2010 | 7/28/2010 | 7/29/2010 | 7/30/2010 | - |
Oil Sands Lab Notebook
This team includes Ann Lesnefsky, Bryce Rajabian, and Kilho Lee.
6/28/2010
Pseudomonas putida KT2440 Antibiotic Resistance
- Protocol for starting overnight culture from -80°C freezer
- All protocols can also be found in the Protocols section of the Notebook
6/29/2010
Pseudomonas putida KT2440 Antibiotic Resistance
6/29/2010 and 6/30/2010
Pseudomonas putida KT2440 Antibiotic Resistance
7/1/2010
Literature Review Napthenic Acids Composition & HPLC Analysis
7/2/2010
Pseudomonas putida KT2440 Antibiotic Resistance
7/7/2010
Ann
Biobrick Transformation with Alex and Jennifer
- Made CaCl2 solution, and 100 mg/mL amphicilin stock solution according to the media section of the wiki
- Started an overnight culture of DH5alpha according to the heat shock transformation protocol
- A 12 mL culture was started because we were multiplying the protocol by 4
7/8/2010
Ann
Biobrick Transformation with Marc, Katie and Audra according to the heat shock transformation protocol
- Started culture for biobrick transformation from over night at 2:30
- At 5:00pm the cultures had overgrown to an OD600 of 1.2
- A a 1:3 dilution of cells was performed and the cells were allowed to go through another doubling period of 20 minutes
- The OD600 was measured again and found to be around 0.500
- the cultures were placed on ice at 5:30 and allowed to chill for 20 minutes
- The washings were performed and the comp cells were resuspended in the residual CaCl2 only
- The OD600 of the comp cells without the glycerol was above 0.3 therefore the comp cells were concentrated enough
- The heat shock was performed for the following biobricks
- BBa_179015
- BBa_179005
- BBa_145001
- BBa_117008
- BBa_117002
- BBa_103006
- The cultures were placed in the 30C shaker to grow up for an hour at 30C at 9:00pm
- The cultures were plated at 10:00pm
7/9/2010
Ann
Biobrick Transformation with Josh, Prae, Charlie according to the miniprep protocol
- After 9 hours of growing at 37C, the plates did not have any colonies on them
- After 22 hours of growth plates that had grown had clear colonies
- The positive and negative controls grew out accordingly
- Only BBa_179015 and BBa_179005 had colonies (both from plate 1 and none from plate 2...)
- BBa_179015 had over 100 colonies
- BBa_179005 had 2 colonies
- 5 mL overnight cultures were started with 100 mg/mL amp at 8pm from a single colony on each plate
7/10/2010
Ann
Biobrick Transformation with Marcus, Bryce, Kilho, Josh, Charlie, Jeremy
Miniprep
- Made frozen stocks from overnight culture of miniprep
- Performed miniprep on BBa_179015 and BBa_179005
- Measured DNA concentration with the Nanodrop
- BBa_179015-11 ng/mL
- BBa_179005-40 ng/mL
Digest
- Ran according to the digest protocol in the protocol section
- Cut parts with EcoRI and PstI
- Digested at 37C for 30 minutes
Gel
- Ran according to the protocol in the protocol section
- Used 8 well comb instead of 13 well comb so the samples were too dilute too see
7/12/2010
Ann
Biobrick Transformation with Jeremy
Gel
- Reran gel from 7/10/2010
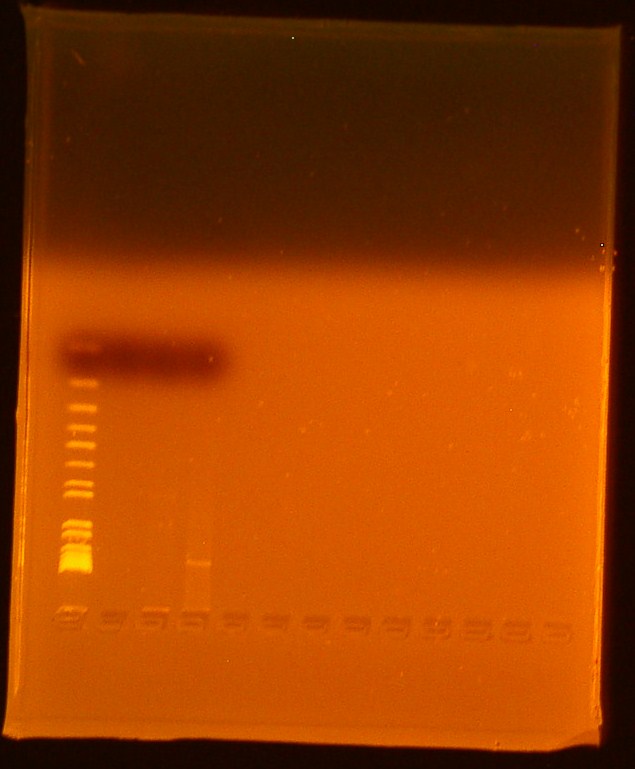
- Lane 1-Invitrogen 1 kb plus ladder
- Lane 2-Digested Bba_179005
- Ran out of undigested Bba_179005
- Lane 3-Digested Bba_179015
- Lane 4-Uncut miniprep plasmid Bba_179015
We found that we successfully extracted BBa_179015, T7-GFP, because there were expected bands at 906 and 2079 as faintly seen in the gel picture above. No appeared for biobrick Bba_179005, T7 promoter, so this biobrick will be transformed again.
Biobrick Transformation take 2 with Jeremy
Autoclaved DI water for transformation and autoclaved sterile containers
- to autoclave sterile containers fill with DI water and decant the water before you start the culture in the container
7/13/2010
Biobrick Transformation take 2 with Jeremy, Marcus, Josh, Kevin, Audra, Katie
Ann
Performed according to the electroporation transformation
- Started the culture at 9:05am
- Removed the culture at 12:05pm with an OD600 of 0.809
- The OD600 of the comp cells were 1.2 after washing
- The time constant for all electroporation were between 5.6 and 5.8 for the following biobricks
- Bba_K117008
- Bba_K117008 #2 (from resuspending remaining part left in registry in 15 uL of ultra pure water)
- Bba_K117002
- Bba_K145001
- Bba_K103006
- Bba_I719005
- The cultures were allowed to grow in the incubator from 2:15-4:00pm
- The cultures started to clump after growing this time
- 100 uL of cells were plated on 100 mg/mL AMP plates
7/14/2010
Biobrick Transformation take 2
Ann
Electroporation Transformation
All of the transformation plates grew out! (minus the negative control of course)
This means we should do all transformations by electroporation from now on. Just check with the Lin Lab to make sure we can use the electroporation machine for a few hours before starting
Miniprep
- Started overnight cultures in 5 mL of LB plus 100 mg/mL AMP
- The following biobricks were started from the transformation plate
- Bba_K117008
- Bba_K117002
- Bba_K145001
- Bba_K103006
- Bba_I719005
- The following biobricks were started from the frozen stock in the -80C freezer
- BBa_K719015
- The following biobricks were started from the transformation plate
INPNC Biobrick part
- We received the INPNC biobrick (Bba_K265008) from UC Davis 2009 team. THANK YOU!!!
- It was shipped on LB plates in a pMA-SK plasmid from Mr. Gene in E. coli DH5alpha
- Since this plasmid has AMP antibiotic resistance I am pouring plates today with Marc and Kevin with 100 mg/mL AMP resistance to streak out the culture tomorrow with Josh
 "
"