Team:HokkaidoU Japan/Notebook/August17
From 2010.igem.org
(Difference between revisions)
(→電気泳動) |
|||
| (8 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{Template:HokkaidoU_Japan}}<div class="linkbar"><div class="prev">[[Team:HokkaidoU_Japan/Notebook/August16|August 16]]</div>[[Team:HokkaidoU_Japan/Notebook|Notebook]]<div class="next">[[Team:HokkaidoU_Japan/Notebook/August18|August 18]]</div></div> | {{Template:HokkaidoU_Japan}}<div class="linkbar"><div class="prev">[[Team:HokkaidoU_Japan/Notebook/August16|August 16]]</div>[[Team:HokkaidoU_Japan/Notebook|Notebook]]<div class="next">[[Team:HokkaidoU_Japan/Notebook/August18|August 18]]</div></div> | ||
| - | = | + | =Gel Extraction= |
| - | == | + | ==Electrophoresis== |
| - | [[Image:HokkaidoU Japan 20100817a.JPG|200px|right|thumb|]] | + | |
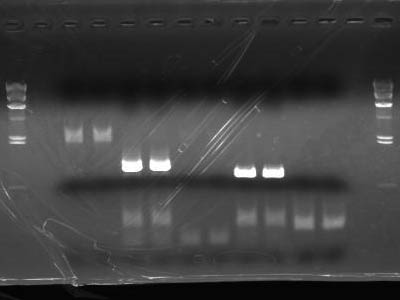
| - | + | [[Image:HokkaidoU Japan 20100817a.JPG|200px|right|thumb|Electrophoresis After Restriction Enzyme Digestion]] | |
| + | |||
| + | 20 uL of DNA digested yesterday was electrophoresed with 4 ul of 6x SB. All acording to the table bellow. | ||
| + | |||
{|class="protocol" | {|class="protocol" | ||
|- | |- | ||
| Line 11: | Line 14: | ||
|- | |- | ||
|1 | |1 | ||
| - | | | + | |Empty |
|- | |- | ||
|2 | |2 | ||
| - | |λ/''Hin''d III | + | |[https://2010.igem.org/Image:HokkaidoU_Pictures_DNA_Marker.png λ/''Hin''d III] |
|- | |- | ||
|3 | |3 | ||
| - | | | + | |Empty |
|- | |- | ||
|4 | |4 | ||
| Line 50: | Line 53: | ||
|- | |- | ||
|14 | |14 | ||
| - | | | + | |Empty |
|- | |- | ||
|15 | |15 | ||
| Line 56: | Line 59: | ||
|- | |- | ||
|16 | |16 | ||
| - | | | + | |Empty |
|- | |- | ||
|17 | |17 | ||
| - | | | + | |Empty |
|} | |} | ||
| - | === | + | ===→[[Team:HokkaidoU_Japan/Protocols|Gel Extraction]]=== |
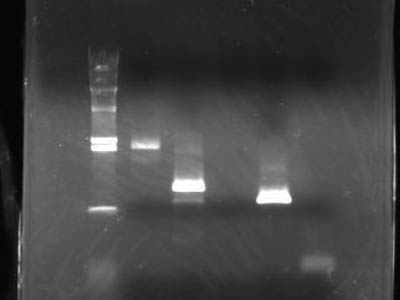
| - | + | [[Image:HokkaidoU Japan 20100817b.jpg|200px|right|thumb|Electrophoresis to measure consentration after gel extraction]] | |
| - | [[Image:HokkaidoU Japan 20100817b.jpg|200px|right|thumb|]] | + | * After ectraction DNA was electrophoresed to check consentration |
| - | * | + | * 2 uL of 6x SB was added to DNA solution of 10 uL |
| - | * DNA solution 10 | + | |
{| class="protocol" | {| class="protocol" | ||
|- | |- | ||
| Line 73: | Line 75: | ||
|- | |- | ||
|1 | |1 | ||
| - | | | + | |Empty |
|- | |- | ||
|2 | |2 | ||
| - | |λ/''Hin''d III | + | |[https://2010.igem.org/Image:HokkaidoU_Pictures_DNA_Marker.png λ/''Hin''d III] |
|- | |- | ||
|3 | |3 | ||
| Line 94: | Line 96: | ||
|- | |- | ||
|8 | |8 | ||
| - | | | + | |Empty |
|} | |} | ||
| - | * | + | * Band of RBS isn't visible |
| - | + | ** This Might due to loss durring extraction | |
| + | →will try again tomorrow | ||
<div style="clear:both"></div> | <div style="clear:both"></div> | ||
| - | =Chloramphenicol | + | =Making of Chloramphenicol LB Medium= |
| - | + | Because we were planing to use pSB1C3 we made LB Medium with chloramphenicol | |
| - | * LB-broth | + | * Added 12.5 g LB-broth and 7.5 g of Agar 7.5 g to 500 mL to autoclaved distiled water, autoclave |
| - | * 500 | + | * Added 500 uL of Chloramphenicol (34 mg/mL) |
| - | * | + | * Poured it to 25 plates, 20 mL per plate |
Latest revision as of 07:36, 27 October 2010
Gel Extraction
Electrophoresis
20 uL of DNA digested yesterday was electrophoresed with 4 ul of 6x SB. All acording to the table bellow.
| Lane | DNA |
| 1 | Empty |
| 2 | λ/Hind III |
| 3 | Empty |
| 4 | pSB1C3/E, P |
| 5 | pSB1C3/E, P |
| 6 | Heat Shock Promotor/E, S |
| 7 | Heat Shock Promotor/E, S |
| 8 | RBS/X, P |
| 9 | RBS/X, P |
| 10 | RFP/E, S |
| 11 | RFP/E, S |
| 12 | double Terminator/E, S |
| 13 | double Terminator/E, S |
| 14 | Empty |
| 15 | λ/Hind III |
| 16 | Empty |
| 17 | Empty |
→Gel Extraction
- After ectraction DNA was electrophoresed to check consentration
- 2 uL of 6x SB was added to DNA solution of 10 uL
| Lane | DNA |
| 1 | Empty |
| 2 | λ/Hind III |
| 3 | Vector |
| 4 | Heat shock promotor |
| 5 | RBS |
| 6 | RFP |
| 7 | double terminator |
| 8 | Empty |
- Band of RBS isn't visible
- This Might due to loss durring extraction
→will try again tomorrow
Making of Chloramphenicol LB Medium
Because we were planing to use pSB1C3 we made LB Medium with chloramphenicol
- Added 12.5 g LB-broth and 7.5 g of Agar 7.5 g to 500 mL to autoclaved distiled water, autoclave
- Added 500 uL of Chloramphenicol (34 mg/mL)
- Poured it to 25 plates, 20 mL per plate
 "
"