Team:Groningen/Hydrophobins
From 2010.igem.org
Chaplins
Biology
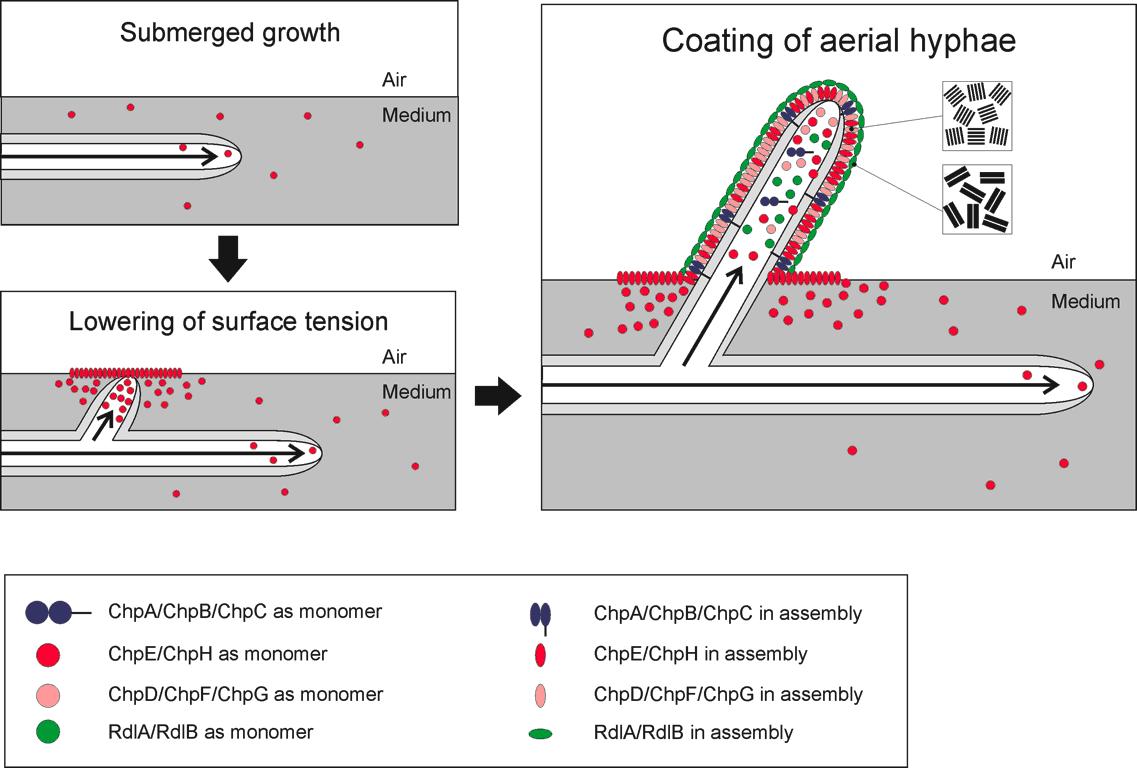
The hydrophobic proteins that we are expressing in our bacillus biofilm are called chaplins and are derived from Streptomyces coelicolor. When Streptomyces is sporulating, hyphae are trying to grow into the air. Since these hyphae are very small, it is very hard for them to break the water-air barrier. This is why Streptomyces produces small hydrophobic proteins on the surface of its hyphae.
Three subgroups
In total there are eight different chaplins. These eight chaplins can be divided into three groups.
Chaplin A-C These chaplins are the largest. They are almost three times the size of the other chaplins (... kD). Besides their size, these chaplins are special because they have a cell wall anchor and a hydrophilic region as well as a hydrophobic region.
Chaplin D and F-H These chaplin are small and only have a hydrophobic region.
Chaplin E This chaplin is also small but it is in vivo released outside the cell to start lowering the watertension before the hyphae actually penetrates the water-air barrier.
Physical properties
Chaplins are interesting because they are amphipathic. This means that they "in theory" can change hydrophilic surfaces into hydrophobic surfaces but in turn can also change hydrophobic surfaces into hydrophilic surfaces.
Chaplins are functional amyloids that will assemble by a catalytic process from monomers in polymer chain forming rod like structure surfaces called amyloid fibers. These fibers are very rigid and hard to break down. These fibers can only be broken when boiled in SDS. They share distinguishing features with the medically important pathogenic amyloid fibers that are characteristic for many neurodegenerative diseases such as Alzheimer's, Huntington's, systemic amyloidosis and the prion diseases.
 "
"