Team:Davidson-MissouriW/MeasuringExpression
From 2010.igem.org
iGEM Davidson – MWSU 2010: Measuring Gene Expression
Insert text here
The system we originally designed to address the knapsack problem had several vital components. The TetA and RFP genes, along with their corresponding gene products, served as our reporters to see if and how a cell solved the knapsack problem. However, we recorded several observations regarding gene expression as we progressed. Characterizing and attempting to understand these foundational problems became one of the new focuses of this team.
Results from past Davidson and Missouri Western research hinted at the presence of a transcriptional terminator within the TetA gene. Any gene located downstream of the 3’ end of the TetA gene did not appear to be expressed. This gene codes for an effluent pump that physically removes the antibody tetracycline from the cell.
To test for the presence of this terminator, we created a construct with a fluorescent protein gene downstream of TetA. If TetA really does block transcription, we would expect to see no fluorescence despite induction of the promoter that precedes the construct (pLac+TetA+RFP). We also tested pLac+TetA+RFP to see if the gene products provided protection from tetracycline compared to a negative control..

The data suggest that the IPTG (0.5mM) inducer decreases cell density in all three cell types. In addition, the construct with TetA before RFP was only able to survive the presence of tetracycline (50ug/mL) when the inducer IPTG was present. These data imply that the inducer did increase gene expression even if overall cell density decreased. The construct fluoresced very little in comparison with the positive control (pLac+RBS+RFP) no matter what the conditions. The data support the hypothesis that there is a transcriptional terminator in the TetA gene.

We performed another battery of tests to compare the pLac+TetA+RFP construct to a construct with the pLac+RFP+TetA orientation. When compared to cells with RFP followed by TetA, the construct’s negligible fluorescence was even more apparent. Interestingly, the cells with the flipped orientation grew poorly in the presence of tetracycline but had a high fluorescence per cell. One possible explanation for this phenomenon is that the fluorescence is divided by such a low number of cells that small variations are magnified. Alternatively, the presence of tetracycline could be selecting for cells with high expression levels of both gene products. These few cells are producing high levels of TetA effluent pumps in order to survive. Correspondingly, they are producing a lot of RFP.

text here

text here

text here
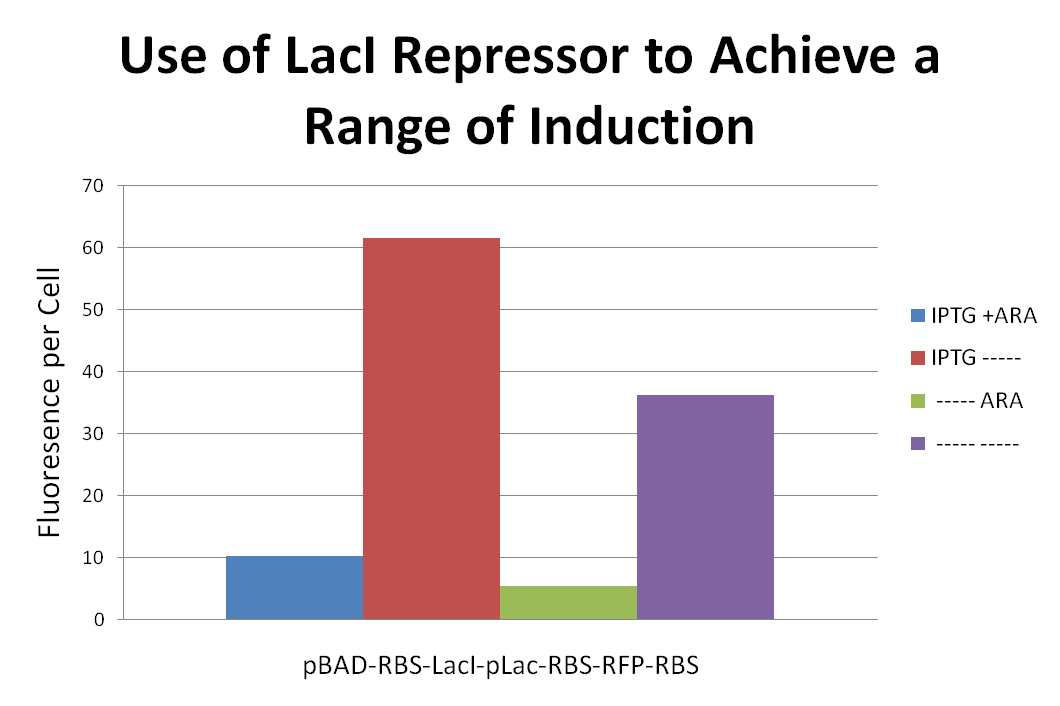
text here
 "
"