Team:Stockholm/Modelling
From 2010.igem.org
(→Bacteria on skin) |
(→Bacteria on skin) |
||
| Line 28: | Line 28: | ||
[http://www.nature.com/jid/journal/v125/n2/full/5603495a.html ] | [http://www.nature.com/jid/journal/v125/n2/full/5603495a.html ] | ||
| + | |||
| + | |||
| Line 53: | Line 55: | ||
[http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002] proposed a mathematical model for lac operon induction in E. Coli. The details that they considered in the model are what we are looking for: external lactose, internal lactose, conversion of lactose to allolactose and glucose, interaction of allolactose with LacI (Lac Repressor) and mRNA. Since the plasmid(?) that we are using to express our genes in bacteria have LacI as repressor, it is reasonable to use the same model as [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002]. | [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002] proposed a mathematical model for lac operon induction in E. Coli. The details that they considered in the model are what we are looking for: external lactose, internal lactose, conversion of lactose to allolactose and glucose, interaction of allolactose with LacI (Lac Repressor) and mRNA. Since the plasmid(?) that we are using to express our genes in bacteria have LacI as repressor, it is reasonable to use the same model as [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002]. | ||
| - | Lac operon is responsible for transport and metabolism of lactose in e coli. It has a promoter site and three structural genes (LacZ, LacY and LacA). Availability of external Lactose and Glucose regulates this operon. In the absence of Lactose, the LacI gene (always expressed) ,which codes for the repressor , represses the expression the of Lac operon. When Lactose is available again for the bacteria, Allolactose (a β-galactosidase side reaction) binds to repressor and makes it impossible for the repressor to bind the operator on Lac operon. This will result in production of high levels of LacZ (β-galactosidase) , LacY (β-galactoside permease) and LacA, and these will lead to more production of Allolactose. Until this point we have the same assumption as [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002]. Here we will introduce the model proposed by [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002] a brief introduction to their model, then we will try to simplify it and | + | Lac operon is responsible for transport and metabolism of lactose in e coli. It has a promoter site and three structural genes (LacZ, LacY and LacA). Availability of external Lactose and Glucose regulates this operon. In the absence of Lactose, the LacI gene (always expressed) ,which codes for the repressor , represses the expression the of Lac operon. When Lactose is available again for the bacteria, Allolactose (a β-galactosidase side reaction) binds to repressor and makes it impossible for the repressor to bind the operator on Lac operon. This will result in production of high levels of LacZ (β-galactosidase) , LacY (β-galactoside permease) and LacA, and these will lead to more production of Allolactose. Until this point we have the same assumption as [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002]. Here we will introduce the model proposed by [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002] a brief introduction to their model, then we will try to simplify it and continue with our gene expression model in bacteria as final stage. |
| + | |||
| + | ==== Yildirim N et al model ==== | ||
| + | |||
| + | In a series of 5 equations, They proposed dynamics for mRNA production, β-galactosidase production, Allolactose, Lactose for Lac operon. In their model they also considered transcriptional and translational delays (ie. β-galactosidase and β-galactoside permease production from mRNA is not instant and takes time). But this model can be simplified. Ahmadzadeh et al. 2005 proposed a simplified model of [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002], where they ignored time delays for transcription and translation. For more simplification they also assumed that β-galactosidase and β-galactoside permease reach their steady state values instantly. | ||
Revision as of 14:01, 21 September 2010
Contents |
Network analysis and prove of concept
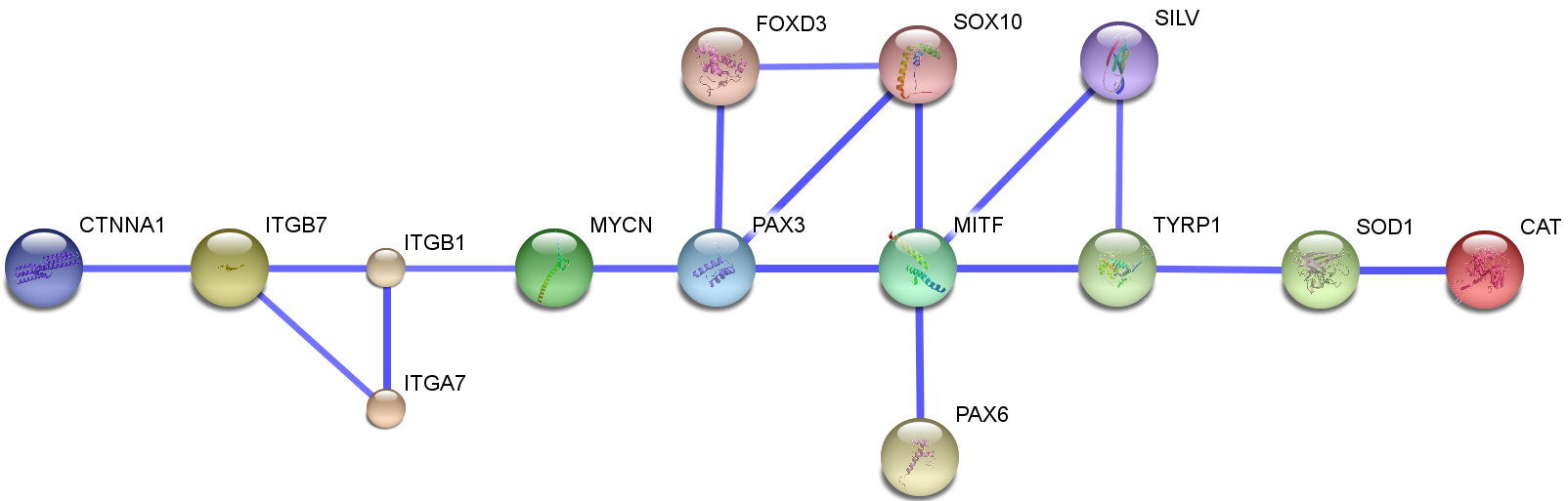
Currently Wet-lab members are working on several proteins that according to previous research have been pointed out to have a positive effect on repigmentation of vitiligo affected skin areas. The idea behind why we chose the biomolecules of interest is presented under the section planning on our wiki homepage. In this section we try to explain a simple interaction network of the specific genes coding the biomolecules. The goal is to give a clear picture of how the genes interact with other potential genes in a way that could result a restroring of the affected skin color.
As a starting point, we began with the article [http://www.ncbi.nlm.nih.gov/pubmed/18426409 Strömberg S et al. (2008)]. In this paper they identified some 859 genes as differentially regulated genes in pigment skin cells called melanocytes. These genes can be classified in several groups based on their possible cellular role (Strömberg et al. 2008). These groups are:
- Developing melanocytes
- Melanin synthesis (melanogenesis) and transport of melanosomes to keratinocytes
- Cell adhesion
- Antigen presenting
In fig. 1, a summarized regulatory network of some candidate genes are illustated. We intentionally chose to present interactions that have currently been investigated in order to support our study.
out of those genes, it seemed that [http://www.ncbi.nlm.nih.gov/gene/4286 MITF] is one of the most regulated and regulating genes in vitiligo disease. This was the first block of the map. . The MITF promoter is targeted by several transcription factors that are important in neural-crest development and signaling. Transcription factors implicated in the regulation of the MITF promoter include PAX3, cAMP-responsive element binding protein (CREB), SOX10, LEF1 (also known as TCF), one cut domain 2 (ONECUT-2) and MITF itself, Fig. 2 (Levy et al., 2006). It also regulates both the survival and differentiation of melanocytes, and enzymes which are necessary for melanin production. (Levy et al., 2006; T.J. Hemesath et al., 1994; N.J. Bentley et al., 1994; K. Yasumoto et al., 1994; C. Bertolotto et al., 1998). So, the regulation of multiple pigmentation and differentiation related genes by MITF (Levy et al., 2006) convinced us that MITF is a central regulator of melanogenesis.
There are evidences for the accumulation of H2O2 in vitiliginous skin (K. U. Schallreuter et al., 1999, 2001, 2006) and low levels of SOD and CAT (A. Jalel et. al., 2008; Koca R et. al., 2004; K. U. Schallreuter et al. 1991; Maresca V et. al. , 1997; ). It was also shown that calcium uptake is defective in vitiliginous skin in keratocytes (K. U. Schallreuter et al. 1988) as well as Melanocytes(K.U. Schallreuter et al., 1996), later the effect of accumulation of H2O2 in the epidermis of patients with vitiligo which leads to disruption calcium homeostasis in the skin was observed (K. U. Schallreuter et al. ,2007). This suggests that an oxidative stress is a pathogenic event in the degeneration of melanocytes (Strömberg S et al. ,2008).
We hypothesized that extra levels of SOD could improve the Melanocytes survivability and help re-pigmentation.
When thinking about re-pigmentation in vitiliginous skin, the first thing comes to mind is lack of melanin. There are evidences that melanocytes are still available in depigmented epidermis of patients with vitiligo even after 25 years (Tobin DJ et. al., 2000). So if melanocytes can't deliver melanin to keratinocytes or they are producing very low amount, it is possible to produce melanin using synthetic biology and deliver it to help re-pigmentation.
[http://www.nature.com/jid/journal/v125/n2/full/5603495a.html ]
Bacteria on skin
Introduction
The challenge that we will face for our bacteria when it comes to the production of proteins, is fusion with cpp (cell penetration peptides). Cause this could lead to the death bacteria as they will penetrate the membrane and eventually kill bacteria when they reach certain concentration. So it is crucial to have the bacteria to begin the production when it comes with contact with skin. We have LacI as repressor, so basically lactose on skin (which is possibly not harmful) will act as inducer. Moreover, a good timing is needed before CPP reaches a critical concentration to kill bacteria. It seems in this scenario we need to take into account possible details such as degradation rates.
Therefore, we are aiming to achieve a predicting model for production of protein in our system of bacteria and have the model to describe production in bacteria from the time bacteria is put on skin until it is dead because of CPPs penetrating bacteria membrane. In the model, we will include production of biomolecules, possible Allolactose/lactose absorption rate by skin and degradation rate in bacteria. However, we will not include or model or try predict re-pigmentation because of high complications in target human cells.
Out of these many details, there are some data we need to know in advance. These details include:
- Concentration of Lactose on skin
- Production rate for biomolecules in host bacteria
- Allolactose/Lactose degradation rate
- Critical concentration of CPP
- Allolactose/Lactose absorption through skin
Choosing proper model
With the above mentioned details, first step should be preparing a proper model for allolactose production, degradation and possible dilution. The reason to this is the binding of allolactose to LacI and inhibiting it from binding to operator, which will result in gene expression.
[http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002] proposed a mathematical model for lac operon induction in E. Coli. The details that they considered in the model are what we are looking for: external lactose, internal lactose, conversion of lactose to allolactose and glucose, interaction of allolactose with LacI (Lac Repressor) and mRNA. Since the plasmid(?) that we are using to express our genes in bacteria have LacI as repressor, it is reasonable to use the same model as [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002].
Lac operon is responsible for transport and metabolism of lactose in e coli. It has a promoter site and three structural genes (LacZ, LacY and LacA). Availability of external Lactose and Glucose regulates this operon. In the absence of Lactose, the LacI gene (always expressed) ,which codes for the repressor , represses the expression the of Lac operon. When Lactose is available again for the bacteria, Allolactose (a β-galactosidase side reaction) binds to repressor and makes it impossible for the repressor to bind the operator on Lac operon. This will result in production of high levels of LacZ (β-galactosidase) , LacY (β-galactoside permease) and LacA, and these will lead to more production of Allolactose. Until this point we have the same assumption as [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002]. Here we will introduce the model proposed by [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002] a brief introduction to their model, then we will try to simplify it and continue with our gene expression model in bacteria as final stage.
Yildirim N et al model
In a series of 5 equations, They proposed dynamics for mRNA production, β-galactosidase production, Allolactose, Lactose for Lac operon. In their model they also considered transcriptional and translational delays (ie. β-galactosidase and β-galactoside permease production from mRNA is not instant and takes time). But this model can be simplified. Ahmadzadeh et al. 2005 proposed a simplified model of [http://www.ncbi.nlm.nih.gov/pubmed/12719218 Yildirim N et al 2002], where they ignored time delays for transcription and translation. For more simplification they also assumed that β-galactosidase and β-galactoside permease reach their steady state values instantly.
 "
"